Did I get this wrong or something but I was thinking that chelated means if the Fe is immediately precipitating out from water or not. That's why I can not use the Flourish Iron.... .
Hi Vazkez,
Chelation is the method by which a valuable chemical is captured and held in handcuffs until that chemical can be transported to it's destination. It is then released at the destination. Normally, the chemical of interest is a metal, which are usually highly reactive. Unfortunately, metals such as Iron, Zinc, Manganese and so forth are so reactive that they have the tendency to react with the first thing that comes along and therefore be lost, or even worse, become toxic. This is the case with Iron. Iron reacts easily with anything. If you have anything made of Iron, you'll know that it will rust very easily, and that's because it's reacting with Oxygen, so you get a precipitated called Iron Oxide - rust. Unfortunately, it is very difficult to separate the Oxygen from the Iron, so basically, the Iron is lost.
The trick of chelation is to combine the metal with another chemical so that this prevents the metal for combining with Oxygen or something else, but that the chemical with which it is combined is not difficult to "unchain" when it get's to the destination.
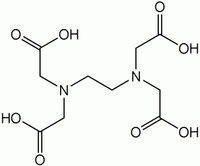
EDTA is a type of acid which acts as a "spiders web" for positively charged metal ions.
It looks like this ordinarily (N is Nitrogen, O is Oxygen, His Hydrogen)
But when it comes into contact with the metal ion, it traps the metal by changing the shape based on the distribution of charges across the structure (the M is the metal ion such as Fe++)
When this complexed structure finds it's way into the plant tissue it is released by the EDTA and is then immediately recaptured by the chelating agents in the plant. These chelates are based on the the chemical called Nicotinamine. You may recognize parts of this chemical's name from cigarettes. Plants generally have loads of this chemical because it's a very effective chelate and there are always toxic metals, which are useful, so they are "sequestered" by the abundant forms of Nicotinamine chelating agents always present in the plant. That's how the plant keeps from poisoning itself and that's why plants are good at removing metals that are dissolved in the water.
The problem with EDTA is that it's attraction to the metals is weakened as the pH of the water approaches neutral (7). It works much better at low pH. Also, hard water interferes with EDTA's ability to hold on to the metal ion. When this bond fails, then the metal ion is released into the water and it immediately reacts with a variety of negatively charged particles in the water. The most highly reactive negatively charged ion is PO4. So, just like your iron bar in air, the two combine to form a compound, which naturally, is called Iron Phosphate, which happens to have very poor solubility in water.
Not every Iron ion reacts with every PO4, but the reaction reduces the total amount of Fe and PO4 that makes it into the plants tissues directly via the leaf.
The thing about Iron though, is that the plants don't really need a lot and in a high tech tank, within an hour or so the plants has as much Fe as it needs, so it really is not a big problem. If you lost 50% of the Iron then just dose more often and it will get there.
The problem in this hobby is that people do not approach Fe rationally. The Fe lovers grossly
overvalue Fe and assume that anything that goes wrong with their plants is due to an Iron deficiency. The Fe haters grossly
undervalue Fe and they always assume that algal blooms are attributable to Fe in the water.
Also if you do not mind could you be so kind and be more specific? Like what can I do to bring my plant to become red? If we do not count light as I think I can not go higher (currently have 106W over 120L).
Well, unfortunately, no one has any idea, other than massive lighting, exactly how to control red in plants. The best that you can do is to ensure high nutrient loading (of all nutrients) in general, and high CO2. As the plants grow closer to the light they will tend to pump out more pigments to protect the leaves. It's not just red, it's all colors because there are many pigment types.
You can also just get plants that are always red, like the various type of Althernanthera or Rotala red.
In some cases, folks were able to get the color changes by using less intense lighting but by changing the spectrum of the lighting to more blue/red content. That's not verified though. You didn't mention what plant this is in any case so it's unclear. There are many reasons for color pigments to appear and they are not all the same reasons for every plant.
Cheers,