Tanksy
Member
I'm "forced" to use RO/DI water for my water changes because my tap water has high NO3 (~40ppm) and I've already bought Seachem Equilibrium to remineralise it.
I've already read many threads about remineralising RO water, but I'm a bit lost. Frankly, the more threads I read, the more confused I am.
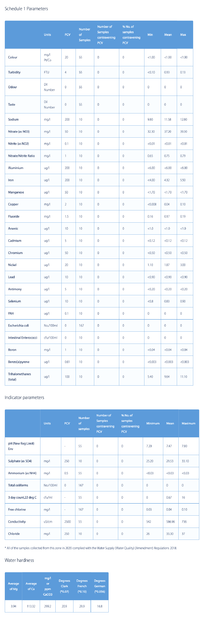
The GH in my tank is 15 dGH. If my maths is correct, to lower it to 9-10 dGH I'd have to add 3 g to 15 litres of RO/DI water (for a 50% WC). Please correct me if I'm wrong.
However, I've just realised that Equilibrium affects only the GH, not the KH. Does that mean that the KH will drop by half if I do 50% WC?
Cheers!
I've already read many threads about remineralising RO water, but I'm a bit lost. Frankly, the more threads I read, the more confused I am.
The GH in my tank is 15 dGH. If my maths is correct, to lower it to 9-10 dGH I'd have to add 3 g to 15 litres of RO/DI water (for a 50% WC). Please correct me if I'm wrong.
However, I've just realised that Equilibrium affects only the GH, not the KH. Does that mean that the KH will drop by half if I do 50% WC?
Cheers!