-
You are viewing the forum as a Guest, please login (you can use your Facebook, Twitter, Google or Microsoft account to login) or register using this link: Log in or Sign Up
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Anyone know where i can get TNC GH Boost?
- Thread starter Kezzab
- Start date
PARAGUAY
Member
You could try email Nigel at TNC for a supplier? Using Equiliberum Seachem but not that cheap.Think Darrell dw1305 and Zeus have advice for making your own
Zeus.
Fertz Meister
I never seen it for sale all the time I've been working on the IFC Calculator, we have always had to use an historical price.
The calculator will clone it without any issues, so for a 100Litre tank doing a 50% WC.

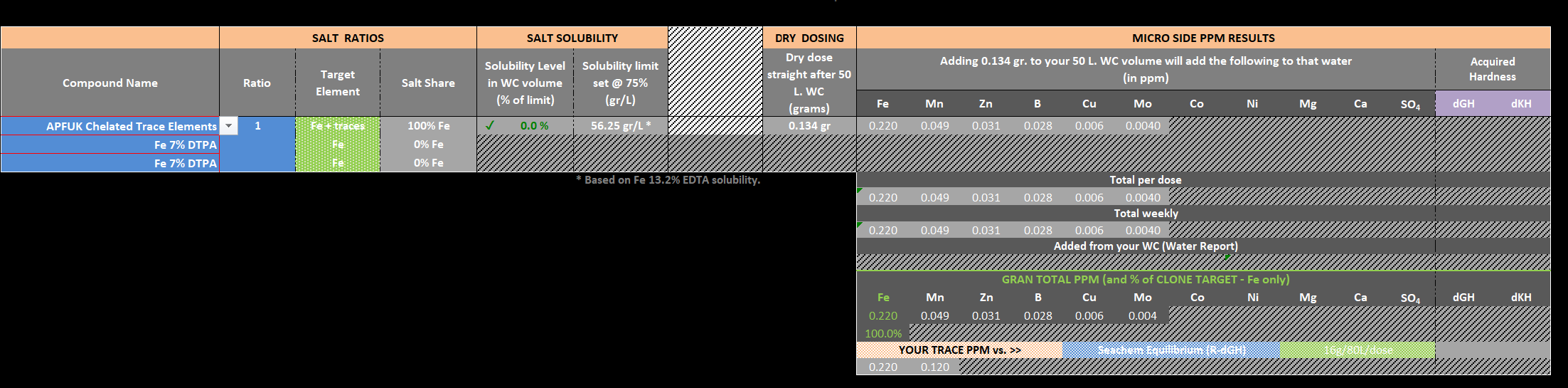
I have been working on a new Remineralising sheet, which has enabled us to clone many more remineralising agents, plus works the Ca:Mg ratio by default

It will be some time before release due to work commitments and moving house.
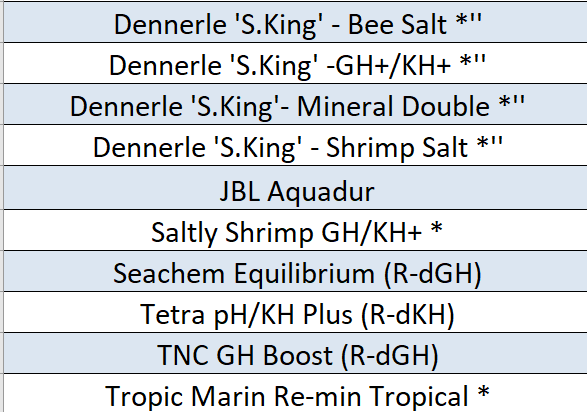
Cloning database so far on new version
Any one after a clone just ask or if the remin agent isn't on list your after let me know as very easy to add
The calculator will clone it without any issues, so for a 100Litre tank doing a 50% WC.
I have been working on a new Remineralising sheet, which has enabled us to clone many more remineralising agents, plus works the Ca:Mg ratio by default
It will be some time before release due to work commitments and moving house.
Cloning database so far on new version
Any one after a clone just ask or if the remin agent isn't on list your after let me know as very easy to add
Zeus.
Fertz Meister
According to our prices it isntWhy not just use CaSO4 and MgSO4? Should be much cheaper.
But depends what your for paying for your salts, you can edit the cost of salts also and calculator does the rest
Plus was balancing out the SO4 : Cl ratio a bit, but I would always go for the cheaper option, unless their is a good reason not too eg why use KNO3 when Urea is cheaper and gives more N - if you need to ask why not use Urea, use KNO3That is what @Zeus. was suggesting with the IFC output above, though with CaCl instead of CaSO4 as it dissolves so easily so is easy to work with.
According to our prices it isnt
View attachment 170634
But depends what your for paying for your salts, you can edit the cost of salts also and calculator does the rest
Plus was balancing out the SO4 : Cl ratio a bit, but I would always go for the cheaper option, unless their is a good reason not too eg why use KNO3 when Urea is cheaper and gives more N - if you need to ask why not use Urea, use KNO3
Is SO4 : CI ratio something people should be considering? This is the first I've heard of it.
These are the ingredients for Seachem's Equilibrium:
Derived from: potassium sulfate, calcium sulfate, magnesium sulfate, ferric sulfate, manganese sulfate.
plantnoobdude
Member
I use calcium chloride dihydrate, and magnesium sulphate heptahydrate, to add only gh. far more cost-effective. and you know what you're putting in your water.
12.5 grams of the calcium chloride dihydrate
and
11.5g magnesium sulphate heptahydrate
4grams of the above remineraliser
Per 20l Water to get 0kh and 6.3 gh
With 31mg/l Ca and 9.6mg/l roughly 3:1 Ca:Mg
12.5 grams of the calcium chloride dihydrate
and
11.5g magnesium sulphate heptahydrate
4grams of the above remineraliser
Per 20l Water to get 0kh and 6.3 gh
With 31mg/l Ca and 9.6mg/l roughly 3:1 Ca:Mg
Zeus.
Fertz Meister
I use calcium chloride dihydrate, and magnesium sulphate heptahydrate, to add only gh. far more cost-effective. and you know what you're putting in your water.
12.5 grams of the calcium chloride dihydrate
and
11.5g magnesium sulphate heptahydrate
4grams of the above remineraliser
Per 20l Water to get 0kh and 6.3 gh
With 31mg/l Ca and 9.6mg/l roughly 3:1 Ca:Mg
Zeus.
Fertz Meister
I don't think so, but some like to reduce the SO4 levels when they canIs SO4 : CI ratio something people should be considering? This is the first I've heard of it.
I don't think so, but some like to reduce the SO4 levels when they can
Is there any reason for this though? Or are they just hedging in case there are some unknown consequences of too much SO4?
Zeus.
Fertz Meister
Or are they just hedging in case there are some unknown consequences of too much SO4?
I think so, it was a something a read at the Barr Report


