Hi all - first time posting. I have a CO2/Ph-related question I'm hoping someone can help me with.
Currently running a relatively heavily planted 60p tank with CO2. It's been up and running for two and a half months. Light from an ADA Aquasky RGB, filter is a Biomaster thermo 250, and I'm dosing daily with 5ml of Tropica specialised with a 50-60% water change each week. There's also a Twinstar M5 in there. Plants appear to be thriving, and other than a touch of green spot on some of the anubias leaves it's algae-free. Here's a pic from a few weeks back:

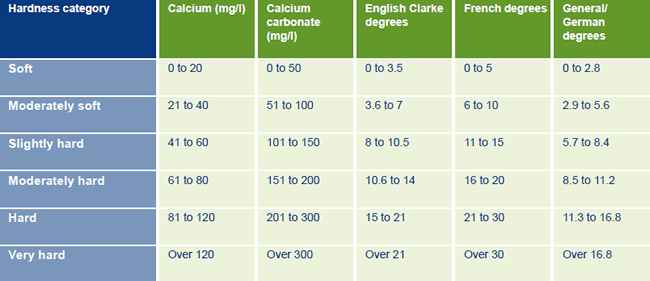
My concern is the Ph level, which seldom drops below 5.7, and just prior to lights-on can dip as low as 5.1. In contrast, my tap water generally hovers around 7.2. Here's some info on the tap water in my area of Cornwall, Falmouth, which South West Water tells me falls under 'moderately soft'. I've not measured my GH or KH.
I'm struggling to understand the disparity between my tap water Ph and that of the tank. Drop checker remains green overnight, so it appears that CO2 simply isn't off-gassing, but there's a reasonable amount of surface agitation and as you can see, I'm using a skimmer.
My question is, how adverse an effect (if any) will this consistently low Ph have on my livestock, which include amano shrimp and horned nerites (I'm sure the ember tetras aren't bothered in the slightest), and what - if anything - should I do to nudge the Ph up a little. Is that even necessary?
Cheers.
Currently running a relatively heavily planted 60p tank with CO2. It's been up and running for two and a half months. Light from an ADA Aquasky RGB, filter is a Biomaster thermo 250, and I'm dosing daily with 5ml of Tropica specialised with a 50-60% water change each week. There's also a Twinstar M5 in there. Plants appear to be thriving, and other than a touch of green spot on some of the anubias leaves it's algae-free. Here's a pic from a few weeks back:
My concern is the Ph level, which seldom drops below 5.7, and just prior to lights-on can dip as low as 5.1. In contrast, my tap water generally hovers around 7.2. Here's some info on the tap water in my area of Cornwall, Falmouth, which South West Water tells me falls under 'moderately soft'. I've not measured my GH or KH.
I'm struggling to understand the disparity between my tap water Ph and that of the tank. Drop checker remains green overnight, so it appears that CO2 simply isn't off-gassing, but there's a reasonable amount of surface agitation and as you can see, I'm using a skimmer.
My question is, how adverse an effect (if any) will this consistently low Ph have on my livestock, which include amano shrimp and horned nerites (I'm sure the ember tetras aren't bothered in the slightest), and what - if anything - should I do to nudge the Ph up a little. Is that even necessary?
Cheers.


