-
You are viewing the forum as a Guest, please login (you can use your Facebook, Twitter, Google or Microsoft account to login) or register using this link: Log in or Sign Up
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Spotless water stations
- Thread starter Ospac
- Start date
Kevin Eades
Member
I don't use it but from the research I did when considering RO water. Its fine for the tank once mineralisedHi guys, has anyone heard of the spotless water stations? I only saw one thread on this but unfortunately it didnt answer my question. Can i use this water is a freshwater tank? Do i need to re-mineralise it?
John q
Member
According to the company website its perfect for aquarium use, and the even claim 0 tds.

 www.spotlesswater.co.uk
www.spotlesswater.co.uk

Ultra Pure Water for Aquariums | Spotless Water
Our water is Ultra Pure which removes pollution and impurities, ensuring the safety of your Aquarium. Our self service stations are accessible across the UK!
MirandaB
Member
Yes it needs to be remineralised as it's pure RO.
I tend to cut mine back with tap water to get the desired parameters.
I tend to cut mine back with tap water to get the desired parameters.
John q
Member
Kevin Eades
Member
Would they not measure it in the pipe straight after it's made?Probably need somebody like @dw1305 to educate us(me) here but I'd assume it would be impossible to produce o tds water if it was exposed to the atmosphere?
John q
Member
Probably yes, but I'd have thought pure water would quickly gobble up carbon, nitrogen e.t.c from the atmosphere. I have been know to be wrong of course 🙃
Either way it sounds like it would be beneficial as ro water.
Either way it sounds like it would be beneficial as ro water.
Hi all,
I was going to say that would only be the tiny proportion of CO2 that goes into solution as H2CO3 and (then H+ and HCO3-) that would go into solution as ions and that you would measure with a conductivity meter.
This is what the <"Veolia ELGA website says">:
cheers Darrel
I think you are right, and that ultrapure water is degassed.it would be impossible to produce o tds water if it was exposed to the atmosphere?
I was going to say that would only be the tiny proportion of CO2 that goes into solution as H2CO3 and (then H+ and HCO3-) that would go into solution as ions and that you would measure with a conductivity meter.
This is what the <"Veolia ELGA website says">:
Because it talks about resistivity, we must be still talking about less than 1 microS conductivity.After Purification
After purification ultrapure water within the purifier has a resistivity of 18.15 MΩ.cm and contains sub-ppb levels of all impurities except dissolved oxygen and nitrogen. As soon as this water is dispensed into a vessel in the laboratory it comes into contact with air. The oxygen and nitrogen contents do not change significantly but CO2 will rapidly be absorbed from the air; hydrogen, bicarbonate and carbonate ions will be produced and the resistivity will fall to about 1.3 MΩ.cm as shown in Figure 1. In normal laboratory practice this is unavoidable. The high conductance of the hydrogen ions means that this change only corresponds to a concentration of 0.5 mg/L CO2 in solution.
cheers Darrel
sparkyweasel
Member
- Joined
- 30 Jun 2011
- Messages
- 2,870
TDS is total dissolved solids, so gasses dissolved from the atmosphere don't count.
Hi all,
You can also get <"undissolved solids"> as well.
cheers Darrel
No, they don't, if you actually measure ppm TDS, but I've never had much joy with <"trying to measure TDS">. In practical terms if you have less than 5 microS (~3 ppm TDS) it is DI water.TDS is total dissolved solids, so gasses dissolved from the atmosphere don't count.
You can also get <"undissolved solids"> as well.
cheers Darrel
John q
Member
Thanks @dw1305 and @sparkyweasel for clarification.
Oldguy
Member
As you said in another thread the only way is to evaporate a known quantity of liquid to dryness and weigh the residue. Still a compromise because the residue has to be 'baked' at a set temperature and for a certain time (depends on the standard that is being used). A right pain to do well.actually measure ppm TDS,
All very academic to our requirements to keep fish and grow plants.
As an aside I wonder about the quality on DI water for car battery maintenance, miss real distilled water.
Hi all,
The advantage of distilled water is that it is always a known entity, because steam distillation doesn't have many moving parts (just the steam really).
We still have some stills plumbed in, (like <"the one below"> ) but we don't use them any more.
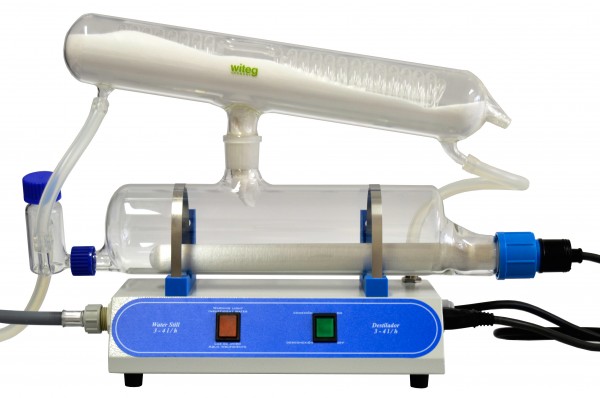
When we used distilled water, rather than DI, the water used to have a slightly higher conductivity , partially because the tap water is very hard (about 18dKH and 650 microS) and you only need very minor contamination to increase the conductivity. We used to get through a lot of acid cleaning the still up every week.
We also used to get through a few stills because the water pressure wasn't very consistent and if pressure dropped enough the cooling water wasn't sufficient to avoid breaking the glass. The other problem is that you need (used to need?) a license to purchase a new distillation unit.
cheers Darrel
While I was answering @Oldguy's question I found the figures for this:but I'd assume it would be impossible to produce o tds water if it was exposed to the atmosphere?
........As distillates are open to the atmosphere before measurements can be made, the value of conductivity is really much less (down to 10 times) than it is really measured. Any ultra pure water in contact with the atmosphere has a conductivity of 1-2 microS/cm and a pH of 5, due to the small amount of CO2 (0.5 ppm) absorbed from the atmosphere.............
A few people have dipped a conductivity meter into RO water they've got from an LFS etc. and found conductivity readings in the hundreds, presumably because the unit wasn't working and nobody had checked. The lab units are <"Veolia ELGA DI units"> and they have all sort of alarms on them should the conductivity rise.As an aside I wonder about the quality on DI water for car battery maintenance, miss real distilled water.
The advantage of distilled water is that it is always a known entity, because steam distillation doesn't have many moving parts (just the steam really).
We still have some stills plumbed in, (like <"the one below"> ) but we don't use them any more.
When we used distilled water, rather than DI, the water used to have a slightly higher conductivity , partially because the tap water is very hard (about 18dKH and 650 microS) and you only need very minor contamination to increase the conductivity. We used to get through a lot of acid cleaning the still up every week.
We also used to get through a few stills because the water pressure wasn't very consistent and if pressure dropped enough the cooling water wasn't sufficient to avoid breaking the glass. The other problem is that you need (used to need?) a license to purchase a new distillation unit.
cheers Darrel
Last edited:
Oldguy
Member
I think that changed with the design of still where the cooling water from the condenser kept the boiler topped up. However red tap is always slow to change. I think in the old days you had an inspection or sent a statement to the Revenue about the still type. Struct me as daft as you could buy Quickfit with no questions asked.a license to purchase a new distillation unit.


