-
You are viewing the forum as a Guest, please login (you can use your Facebook, Twitter, Google or Microsoft account to login) or register using this link: Log in or Sign Up
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
surface agitation affect liquid c02
- Thread starter Aqua Hero
- Start date
-
- Tags
- c02 liquid co2
parotet
Member
No, surface agitation degasses the co2 you try to dissolve, while liquid co2 is not a gas but a chemical compound that can play a similar role co2 plays during the photosynthesis
Jordi
Jordi
AndyMcD
Member
The Flourish Excel FAQs confirms this (http://www.seachem.com/support/FAQs/FlourishExcel.html).
As liquid carbon isn't a gas, surface agitation doesn't appear to affect concentration.
I'm trying to design a planted tank for fish from faster flowing streams that need higher levels of oxygen all of the time.
Aside from photosynthesis, surface agitation seems to be the best way of getting oxygen into the water. However, surface agitation will degas CO2.
Switching an air stone on after lights out means you can get oxygen in when the plants are not photosynthesising. Howeve, high swings of CO2 level appears to increase BBA.
Also, to achieve required CO2 concentration at lights on, the air stone would have to be switched off two hours before. For two hours there would be little oxygen coming into the system from either the air stone or the plants and increasing CO2 levels. Not good for the fish.
Therefore, I'm planning to have surface agitation, adding liquid carbon and selecting plants that Tropica (or other suppliers) state require a lower demand for CO2.
Not sure if this helps. This doesn't seem to be a common approach.
As liquid carbon isn't a gas, surface agitation doesn't appear to affect concentration.
I'm trying to design a planted tank for fish from faster flowing streams that need higher levels of oxygen all of the time.
Aside from photosynthesis, surface agitation seems to be the best way of getting oxygen into the water. However, surface agitation will degas CO2.
Switching an air stone on after lights out means you can get oxygen in when the plants are not photosynthesising. Howeve, high swings of CO2 level appears to increase BBA.
Also, to achieve required CO2 concentration at lights on, the air stone would have to be switched off two hours before. For two hours there would be little oxygen coming into the system from either the air stone or the plants and increasing CO2 levels. Not good for the fish.
Therefore, I'm planning to have surface agitation, adding liquid carbon and selecting plants that Tropica (or other suppliers) state require a lower demand for CO2.
Not sure if this helps. This doesn't seem to be a common approach.
- Joined
- 8 May 2015
- Messages
- 278
Nope it was very helpful considering all my plants have low demands in c02The Flourish Excel FAQs confirms this (http://www.seachem.com/support/FAQs/FlourishExcel.html).
As liquid carbon isn't a gas, surface agitation doesn't appear to affect concentration.
I'm trying to design a planted tank for fish from faster flowing streams that need higher levels of oxygen all of the time.
Aside from photosynthesis, surface agitation seems to be the best way of getting oxygen into the water. However, surface agitation will degas CO2.
Switching an air stone on after lights out means you can get oxygen in when the plants are not photosynthesising. Howeve, high swings of CO2 level appears to increase BBA.
Also, to achieve required CO2 concentration at lights on, the air stone would have to be switched off two hours before. For two hours there would be little oxygen coming into the system from either the air stone or the plants and increasing CO2 levels. Not good for the fish.
Therefore, I'm planning to have surface agitation, adding liquid carbon and selecting plants that Tropica (or other suppliers) state require a lower demand for CO2.
Not sure if this helps. This doesn't seem to be a common approach.
Sent from my D2303 using Tapatalk
- Joined
- 8 May 2015
- Messages
- 278
Thank youNo, surface agitation degasses the co2 you try to dissolve, while liquid co2 is not a gas but a chemical compound that can play a similar role co2 plays during the photosynthesis
Jordi
Sent from my D2303 using Tapatalk
Hi all,
Systems with a large gas exchange system (via a wet and dry trickle filter, a large surface area to volume tank or a linear flow) will maintain higher CO2 levels than other low tech systems when the plants are actively photosynthesizing.
cheer Darrel
If you have high levels of flow you will maintain CO2 and O2 levels close to their equilibrium values with the atmosphere. I recommend this approach, partially because I like the tank to be well oxygenated at all times.I'm planning to have surface agitation, adding liquid carbon and selecting plants that Tropica (or other suppliers) state require a lower demand for CO2.
Systems with a large gas exchange system (via a wet and dry trickle filter, a large surface area to volume tank or a linear flow) will maintain higher CO2 levels than other low tech systems when the plants are actively photosynthesizing.
cheer Darrel
Systems with a large gas exchange system (via a wet and dry trickle filter, a large surface area to volume tank or a linear flow) will maintain higher CO2 levels than other low tech systems when the plants are actively photosynthesizing.
Any ideas what are the maximum ppms of co2 that we can maintain just via agiatation Darrel? I understand T and P play a big role here.
Only when lights are on. Degassing CO2 via an airstone when lights are off and starting to add CO2 before lights are on does not cause BBA.Howeve, high swings of CO2 level appears to increase BBA.
3ppm I read somewhere.Any ideas what are the maximum ppms of co2 that we can maintain just via agiatation
Hi all,
Looking at the temperature solubility curve from <http://www.engineeringtoolbox.com/gases-solubility-water-d_1148.html>.
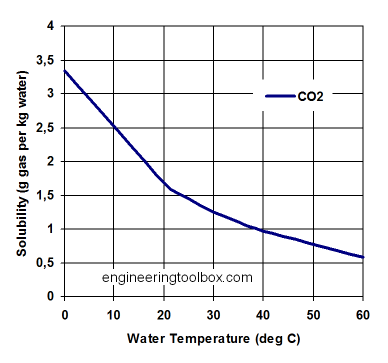
To get from the curve to ppm of CO2 in water is beyond my maths, but I'll assume it is close to the 3ppm quoted in the literature.
cheers Darrel
Temperature is relevant, but you can largely ignore air pressure and the conductivity of the water, unless you are a marine aquarist and live in Denver.I understand T and P play a big role here.
I don't have any figures specifically for CO2, because with polluted water you are mainly trying to out-gas the CO2, but if you look at oxygen, you can maintain it pretty close to saturation, even with larger BOD values than you would get in an aquarium.Any ideas what are the maximum ppms of co2 that we can maintain just via agiatation Darrel?I
Looking at the temperature solubility curve from <http://www.engineeringtoolbox.com/gases-solubility-water-d_1148.html>.
To get from the curve to ppm of CO2 in water is beyond my maths, but I'll assume it is close to the 3ppm quoted in the literature.
cheers Darrel
Here is a link on how to calculate the amount of CO2 from the atmosphere that dissolves into water using Henry's law.
butane.chem.uiuc.edu/pshapley/GenChem1/L23/web-L23.pdf
However the answer it gives is 4.25 x 10-7 M. Some one needs to convert this the ppm.
butane.chem.uiuc.edu/pshapley/GenChem1/L23/web-L23.pdf
However the answer it gives is 4.25 x 10-7 M. Some one needs to convert this the ppm.
However the answer it gives is 4.25 x 10-7 M. Some one needs to convert this the ppm.
To change into ppm we do the following:
4.25 x10-7 mol of co2/l x 44 g of CO2/mol of co2 x 1000mg CO2/g CO2 = 0.0187 mg CO2/l water= 0.0187 ppm
Whats wrong here?
Last edited:
Hi all,
Looking at the paper I think the equation you need is:
[CO2] = P/KH = 3.87 x 10-4 atm/29.41 atm M-1= 1.32 x 10-5M,
Which gives you 0.58ppm CO2 in pure H2O.
0.58 = 0.0000132 x 44,000, (where 44,000 ppm = 1 mol. CO2).
I assume there must be some other factors to raise this to 3ppm CO2.
cheers Darrel
Looking at the paper I think the equation you need is:
[CO2] = P/KH = 3.87 x 10-4 atm/29.41 atm M-1= 1.32 x 10-5M,
Which gives you 0.58ppm CO2 in pure H2O.
0.58 = 0.0000132 x 44,000, (where 44,000 ppm = 1 mol. CO2).
I assume there must be some other factors to raise this to 3ppm CO2.
cheers Darrel
xim
Member
- Joined
- 19 Mar 2010
- Messages
- 499
2-3 PPM vs 0.5 PPM...
I think the 2-3 PPM info is probably from http://www.hallman.org/plant/booth2.html .
Although the measurement was done by using LaMotte's test kits, the margin of error of 2 PPM
and the resolution of 1 PPM might be too coarse for this measurement. And it's an aquarium,
there must be some CO2 producing activities in it.
All other people and documents I've found state ~0.5 PPM.
I think the 2-3 PPM info is probably from http://www.hallman.org/plant/booth2.html .
Although the measurement was done by using LaMotte's test kits, the margin of error of 2 PPM
and the resolution of 1 PPM might be too coarse for this measurement. And it's an aquarium,
there must be some CO2 producing activities in it.
All other people and documents I've found state ~0.5 PPM.
AndyMcD
Member
If you wished to increase the carbon available to the plants above ~0.5ppm in a tank with surface agitation, would the best next step be to add liquid carbon rather than gaseous CO2?
I'm assuming that the answer is liquid carbon, as adding gaseous CO2 will be lost through the agitated surface.
Will increasing the level of oxygen also speed up the breakdown of organic material, leading to the production of CO2 (although in the same way is this likely to be lost due to surface agitation)?
I'm sorry, I'm very inexperienced, but I'm trying to convince myself that with rheoliphic fish investing in CO2 isn't appropriate. Sticking with liquid carbon may be the better option.
I'm assuming that the answer is liquid carbon, as adding gaseous CO2 will be lost through the agitated surface.
Will increasing the level of oxygen also speed up the breakdown of organic material, leading to the production of CO2 (although in the same way is this likely to be lost due to surface agitation)?
I'm sorry, I'm very inexperienced, but I'm trying to convince myself that with rheoliphic fish investing in CO2 isn't appropriate. Sticking with liquid carbon may be the better option.
xim
Member
- Joined
- 19 Mar 2010
- Messages
- 499
This is the first time I hear about “rheoliphic” fish. Google also doesn't help much. So I have no idea about this fish and CO2. But generally, smaller fish are more CO2 tolerant than larger fish.
Liquid carbon is not also very safe for animals. It is a diluted solution of a disinfectant (glutaraldehyde) used in hospitals. Overdosing can kill both plants and animals. But the dosage is much easier to assess than CO2, so may be it could be said it's safer because of that.
Liquid carbon is not also very safe for animals. It is a diluted solution of a disinfectant (glutaraldehyde) used in hospitals. Overdosing can kill both plants and animals. But the dosage is much easier to assess than CO2, so may be it could be said it's safer because of that.
AndyMcD
Member
I am so embarrassed.
I meant rheophilic - river loving.
Serves me right for trying to use big words!
I meant rheophilic - river loving.
Serves me right for trying to use big words!
Alexander Belchenko
Member
2-3 PPM vs 0.5 PPM...
I think the 2-3 PPM info is probably from http://www.hallman.org/plant/booth2.html .
Although the measurement was done by using LaMotte's test kits, the margin of error of 2 PPM
and the resolution of 1 PPM might be too coarse for this measurement. And it's an aquarium,
there must be some CO2 producing activities in it.
All other people and documents I've found state ~0.5 PPM.
Interesting reading on the link you have provided. Esp. caught my attention this sentence and note in brackets:
At the end of most of the tests, CO2 again measured about 2-3 ppm, indicating that this was the equilibrium value for the experimental conditions (note that the altitude was 5000 feet above sea level).
I don't remember exactly how solubility of oxygen/CO2 is changed with altitude though, but IIRC with higher altitude pressure is decreasing and therefore solubility should decrease too.
I don't remember exactly how solubility of oxygen/CO2 is changed with altitude though, but IIRC with higher altitude pressure is decreasing and therefore solubility should decrease too.
Yes, I would say the higher the altitude, the less co2 dissolved because there is less partial pressure from the co2. So I think youre right A.B.


