ElleDee
Member
Here's a table from my water report. There is no context given for any of this, it's just a standalone table nestled in pages of stuff about contaminant testing.
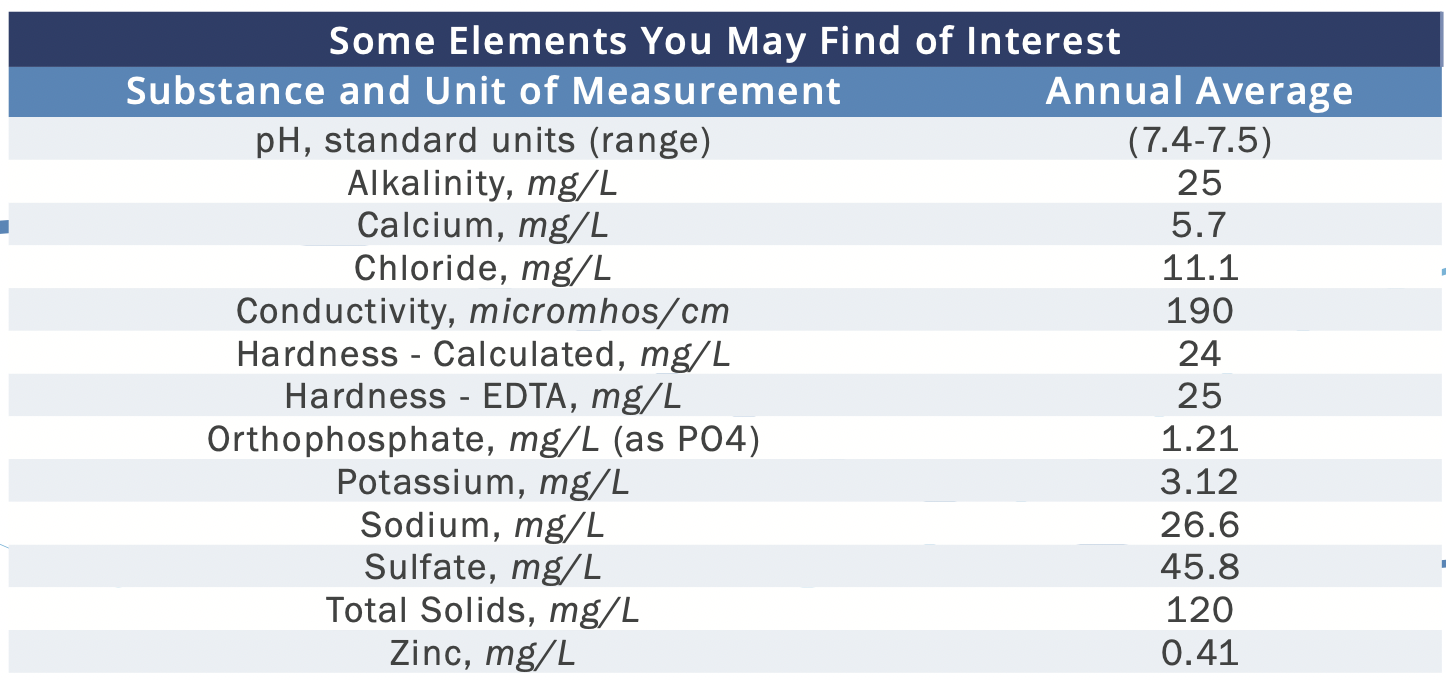
Ok, so I know most of the alkalinity in aquariums is from carbonate, is that also the case here?
And "Hardness" here is GH?
So if 5.7 ppm of Ca2+ = 14. 3 ppm GH, then the remaining 9.7 ppm GH = ~2.4 ppm Mg2+ ...right?
I swear I know the difference between GH and KH, but when everyone uses different terminology I start second guessing myself.
Ok, so I know most of the alkalinity in aquariums is from carbonate, is that also the case here?
And "Hardness" here is GH?
So if 5.7 ppm of Ca2+ = 14. 3 ppm GH, then the remaining 9.7 ppm GH = ~2.4 ppm Mg2+ ...right?
I swear I know the difference between GH and KH, but when everyone uses different terminology I start second guessing myself.


