-
You are viewing the forum as a Guest, please login (you can use your Facebook, Twitter, Google or Microsoft account to login) or register using this link: Log in or Sign Up
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Excess of K, Na, Ca, Mg... fact or myth?
- Thread starter gmartins
- Start date
For aquatic plants this is a myth. For terrestrial this may have some validity.
Cheers,
Cheers,
niru
Member
Why Clive, why?? 

+1Why Clive, why??
Well, I mean there are some major differences between a terrestrial versus aquatic environment (which shouldn't be Earth shattering news). The K+ ion is not integrated into organic structures in a plant. For example, Nitrogen is embedded into amino acids, enzymes and proteins. Carbon is embedded into the structure of membranes, tissues and carbohydrates. Phosphorous is embedded into DNA, ATP and so forth. But K+ lives within the "bloodstream" of the plant. It is not combined into molecules, but exists in a soluble form in the cytoplasm of cells and in other liquids. K+ has so many functions, but some of those functions are the electrochemical properties which are similar to salinity. So in a terrestrial plant the movement of K+ into and out of fluids has an effect on cell turgidity, so these plants can suffer symptoms similar to "salt stress" or Sodium (Na+) toxicity. In an aquatic environment though this is not a big deal because K+ can move out of the tissues and leech back into the water column by simple osmosis. K+ is probably THE most mobile of the nutrients, so it is much more easily regulated in aquatics than in terrestrials.
You know, a lot of people complain about K+ stress causing symptoms manifest in Ca++ blocking, but this is rubbish because they misdiagnose. One of the symptoms of terrestrial plants Ca++ shortage is stunted leaf tips, or curled leaves, but in submersed plants this same symptom is actually caused by poor CO2. So their analysis is a combination of poor diagnosis plus misapplied information.
Besides, I've tried many times to do a K+ overdose and I'm not able to verify it at all. Of course I haven't tried it on every plant, so I can't say unilaterally, but in the 100 or so popular varieties I have grown I haven't seen any signs of toxicity while deliberately pushing the K+ toxicity envelope.
Here is P. stelleta at 100+ ppm K+ easily accomplished by adding copious amounts of KNO3 and KH2PO4.

Here is L. aromatica pushed to 130+ppm K+. This is almost 5X EI dosing. I see no evidence of curled plant leaves. If your leaves are deformed then this means you have too much light and are not adding enough CO2. But people don't want to hear that because getting enough CO2 into the tank is a tiresome process, so they would rather hear tales of exotic toxicity syndromes. So if you want to say that excess K+ is a problem, you cannot say that K+ alone causes problems. The argument must be modified to be: "excessive K+ in the presence of some other factor" causes problems.

Cheers,
You know, a lot of people complain about K+ stress causing symptoms manifest in Ca++ blocking, but this is rubbish because they misdiagnose. One of the symptoms of terrestrial plants Ca++ shortage is stunted leaf tips, or curled leaves, but in submersed plants this same symptom is actually caused by poor CO2. So their analysis is a combination of poor diagnosis plus misapplied information.
Besides, I've tried many times to do a K+ overdose and I'm not able to verify it at all. Of course I haven't tried it on every plant, so I can't say unilaterally, but in the 100 or so popular varieties I have grown I haven't seen any signs of toxicity while deliberately pushing the K+ toxicity envelope.
Here is P. stelleta at 100+ ppm K+ easily accomplished by adding copious amounts of KNO3 and KH2PO4.

Here is L. aromatica pushed to 130+ppm K+. This is almost 5X EI dosing. I see no evidence of curled plant leaves. If your leaves are deformed then this means you have too much light and are not adding enough CO2. But people don't want to hear that because getting enough CO2 into the tank is a tiresome process, so they would rather hear tales of exotic toxicity syndromes. So if you want to say that excess K+ is a problem, you cannot say that K+ alone causes problems. The argument must be modified to be: "excessive K+ in the presence of some other factor" causes problems.

Cheers,
NatureBoy
Member
- Joined
- 27 Aug 2008
- Messages
- 374
I agree that the vast majority of aquatic plants flourish without any adverse regulatory issues regarding K+ concentrations in the water column, certainly true of the Pogostemon I've had.
However certain plants, perhaps those adapted to water with extremely low "hardwater" ion concentrations may not be able to regulate the K+ ion so efficiently. Plants that come to mind where I've seen this most apparent are L arcuata, R macaranda - when I lay off the K+, normal leaf formation, all other factors constant (as possible😉)
I don't suggest this simply to be contentious, but observing growth under different concentrations of nutrients / lights / CO2 has led me to this opinion.
I'm not a potassium "hater", rather a potassium respecter.
However certain plants, perhaps those adapted to water with extremely low "hardwater" ion concentrations may not be able to regulate the K+ ion so efficiently. Plants that come to mind where I've seen this most apparent are L arcuata, R macaranda - when I lay off the K+, normal leaf formation, all other factors constant (as possible😉)
I don't suggest this simply to be contentious, but observing growth under different concentrations of nutrients / lights / CO2 has led me to this opinion.
I'm not a potassium "hater", rather a potassium respecter.
zanguli-ya-zamba
Seedling
Thanks for the explanation Clive !!
As usual when you have to explain us really scientific matters you make it understandable !
Thank you for that !
By the way I am reading all the Barr reports since few days, and I am really happy that I have took the decision to start this long journey of reading !
Regards
Sent from my iPhone using Tapatalk
As usual when you have to explain us really scientific matters you make it understandable !
Thank you for that !
By the way I am reading all the Barr reports since few days, and I am really happy that I have took the decision to start this long journey of reading !
Regards
Sent from my iPhone using Tapatalk
Thanks for your in depth reply Clive.
I thought it was a myth myself but it was worth asking anyways... This question was put by a friend who has copious amounts of light, CO2 and ferts so it is hard to say what was hard to say upfront what the problem might be. Despite the yellow drop-checker, it could well be that the amount of co2 is not sufficient given the high lights for that particular species.
cheers
GM
I thought it was a myth myself but it was worth asking anyways... This question was put by a friend who has copious amounts of light, CO2 and ferts so it is hard to say what was hard to say upfront what the problem might be. Despite the yellow drop-checker, it could well be that the amount of co2 is not sufficient given the high lights for that particular species.
cheers
GM
Hi zanguli,
Yeah keep reading mate. Lots of great information there. Be sure to read every single one of his pdf files in the Newsletter section.
Yes, although I may seem recalcitrant, I do have an open mind about it, and your observations could point to the truth. What happens though in the hobby is that unsubstantiated information somehow gets legitimized because it seems easier to swallow than the truth. Somehow, the exceptions become the rule. I agree that there may be some issues with certain highly specialized plants, but how many exclusively soft water plants are there in comparison to the total number of available plants? A handful, really, and yet folks tend to extrapolate the problems associated with this small minority to all other species, so that any report of curled leaves are immediately attributed to K+ toxicity instead of approaching the problem in a more rational and systematic fashion.
As you mention, control is a very difficult proposition. This tunnel vision that people tend to have extends even to the point where someone will make two or more changes, will see an improvement, and will automatically attribute the improvement to their favorite factor and will ignore the reality that several things were changed. So the troubleshooting is reduced to guesswork and luck. I'm not implying that you are guilty of this, only that it does happen frequently and the reader has no way of verifying. For example, have you thought about actually setting up several small tanks as closely matched as possible and trying to keep everything the same, except for the K+ levels to see what the response is comparatively? That is really the only way to verify. Also, it must be repeatable. After having solved the problem as you mentioned, did you then add K+ to see if the leaves returned to their distorted condition?
If you have a look at Tom Barr's "Dutch something or the other" 120 Gal | UK Aquatic Plant Society you'll see that he doses the tank with a fairly standard EI scheme and does not suffer this issue. That tank is a revolving door, with species coming and going but R. macandra and L. acuarta have found their way into that tank without issues. According to Tom, KH is much more of an issue with these soft water species than K+ or GH, so it's something worth looking into.
Cheers,
Yeah keep reading mate. Lots of great information there. Be sure to read every single one of his pdf files in the Newsletter section.
Hi,I agree that the vast majority of aquatic plants flourish without any adverse regulatory issues regarding K+ concentrations in the water column, certainly true of the Pogostemon I've had.
However certain plants, perhaps those adapted to water with extremely low "hardwater" ion concentrations may not be able to regulate the K+ ion so efficiently. Plants that come to mind where I've seen this most apparent are L arcuata, R macaranda - when I lay off the K+, normal leaf formation, all other factors constant (as possible😉)
I don't suggest this simply to be contentious, but observing growth under different concentrations of nutrients / lights / CO2 has led me to this opinion.
I'm not a potassium "hater", rather a potassium respecter.
Yes, although I may seem recalcitrant, I do have an open mind about it, and your observations could point to the truth. What happens though in the hobby is that unsubstantiated information somehow gets legitimized because it seems easier to swallow than the truth. Somehow, the exceptions become the rule. I agree that there may be some issues with certain highly specialized plants, but how many exclusively soft water plants are there in comparison to the total number of available plants? A handful, really, and yet folks tend to extrapolate the problems associated with this small minority to all other species, so that any report of curled leaves are immediately attributed to K+ toxicity instead of approaching the problem in a more rational and systematic fashion.
As you mention, control is a very difficult proposition. This tunnel vision that people tend to have extends even to the point where someone will make two or more changes, will see an improvement, and will automatically attribute the improvement to their favorite factor and will ignore the reality that several things were changed. So the troubleshooting is reduced to guesswork and luck. I'm not implying that you are guilty of this, only that it does happen frequently and the reader has no way of verifying. For example, have you thought about actually setting up several small tanks as closely matched as possible and trying to keep everything the same, except for the K+ levels to see what the response is comparatively? That is really the only way to verify. Also, it must be repeatable. After having solved the problem as you mentioned, did you then add K+ to see if the leaves returned to their distorted condition?
If you have a look at Tom Barr's "Dutch something or the other" 120 Gal | UK Aquatic Plant Society you'll see that he doses the tank with a fairly standard EI scheme and does not suffer this issue. That tank is a revolving door, with species coming and going but R. macandra and L. acuarta have found their way into that tank without issues. According to Tom, KH is much more of an issue with these soft water species than K+ or GH, so it's something worth looking into.
Cheers,
zanguli-ya-zamba
Seedling
That's what I am reading right now hehe !
Sent from my iPhone using Tapatalk
Sent from my iPhone using Tapatalk
NatureBoy
Member
- Joined
- 27 Aug 2008
- Messages
- 374
Hi
Yes I've read Tom's journal, followed lots of his threads / responses and respect most high his contribution for our hobby! Regarding the difficulties of growing certain plants his conclusion is that KH is the most probable cause of difficulty for people raising this subset of plants.
Now he lives in a place with low KH water and you can't argue with his success with these "difficult" / fussy plants...
I use tapwater labeled "moderately hard" with a higher KH. It's fair to predict that I struggle to maintain sufficient / steady dissolved carbon dioxide with that wall of carbonate getting in the way and as a result the growth from time to time would be anticipated to waiver - hence crinkly new growth from time to time. This would be most prominent with increasing biomass and flow restrictions. I'm pretty sure that will happen, so make every effort to keep CO2 as optimum as possible and indeed all plants are actively photosynthesizing throughout the photoperiod without showing signs of deficiencies. On this basis I am fairly certain I've covered the CO2 needs of my water column.
It's to this happy mix that I'm fairly confident I can distort growth in L arcuata fairly rapidly if I up the K....it ain't scientific but it's the best I can do with one tank.
This is an interesting thread...Dosing with Ammonia and Urea | UK Aquatic Plant Society the suspect in this thread is Nitrate, where some pretty notable people like JamesC were noticing stunting and then an improvement when switching from potassium nitrate to urea...what they didn't twig was that it was just as probable that it was the K+ of KNO3 rather than the nitrate that was the source of their woes, that when switched for urea was taking the K+ down.
Their is lots of this type of crinkly leaf / deformed new growth observations throughout the forums, are people happy that the situation has been resolved?
cheers
Yes I've read Tom's journal, followed lots of his threads / responses and respect most high his contribution for our hobby! Regarding the difficulties of growing certain plants his conclusion is that KH is the most probable cause of difficulty for people raising this subset of plants.
Now he lives in a place with low KH water and you can't argue with his success with these "difficult" / fussy plants...
I use tapwater labeled "moderately hard" with a higher KH. It's fair to predict that I struggle to maintain sufficient / steady dissolved carbon dioxide with that wall of carbonate getting in the way and as a result the growth from time to time would be anticipated to waiver - hence crinkly new growth from time to time. This would be most prominent with increasing biomass and flow restrictions. I'm pretty sure that will happen, so make every effort to keep CO2 as optimum as possible and indeed all plants are actively photosynthesizing throughout the photoperiod without showing signs of deficiencies. On this basis I am fairly certain I've covered the CO2 needs of my water column.
It's to this happy mix that I'm fairly confident I can distort growth in L arcuata fairly rapidly if I up the K....it ain't scientific but it's the best I can do with one tank.
This is an interesting thread...Dosing with Ammonia and Urea | UK Aquatic Plant Society the suspect in this thread is Nitrate, where some pretty notable people like JamesC were noticing stunting and then an improvement when switching from potassium nitrate to urea...what they didn't twig was that it was just as probable that it was the K+ of KNO3 rather than the nitrate that was the source of their woes, that when switched for urea was taking the K+ down.
Their is lots of this type of crinkly leaf / deformed new growth observations throughout the forums, are people happy that the situation has been resolved?
cheers
Hi,
The carbonate hardness of the water has no bearing on the solubility of gasses in the water, therefore there is no difference in injecting CO2 in the high KH water as opposed to injecting CO2 in low KH water. This is another fallacy that is prevalent throughout the hobby. The solubility of CO2 has nothing to do with alkalinity and nothing to do with carbonate hardness. CO2 solubility in fresh water is strictly limited to the temperature and pressure. Alkalinity and carbonate hardness are not the same thing, and you KH test kit does not measure carbonate hardness, it measure alkalinity, which might be affected by this ion.
If you noticed in Tobi's thread there are those who report increased performance as well as those who report little to no improvement. Again, because many people lack the control as well as lack the reporting skills, it's very difficult to ascertain WHY there is a difference in performance. We need to understand the WHY and that will give us a clue. We do not know for example whether the reporter's tank was previously under-dosed with NO3 and whether the tank was overly lit. Adding urea also produces CO2 as it is converted to NH3->NH4. It could also be that the reporting hobbyist had water that was low in magnesium or that they were using RO without remineralizing and that the simple addition of the Magnesium Nitrate addressed this. NH4 also delivers about 4 times the level of Nitrogen than NO3 does, so of course we should expect an improvement when adding more N per unit mass.
So handing out a sample recipe without any control whatsoever and having people report on results is not really a sound method of collecting data, and drawing a conclusion based on this data is not sound either.
Again, I'm not saying that there is no possibility of excess K+ having a negative effect in certain plants under certain conditions. What I'm saying is that we need better testing to verify the claims and that we should not limit our conclusions to only excess K+ when there are clear examples that fixing the CO2 and distribution, which everyone erroneously assumes is easy, clearly has shown to fix the issue in many cases. Additionally, there are examples of these same plants being grown in high K+ tanks which do not suffer the deformation. Plants have been grown using the ADA system via ADA Aquasoil with the addition of "Brighty K". New ADA aquasoil has about 100X EI levels of NPK, and, Brighty K is a K+ supplement. Viktorlantis, for example is able to grow these plants without any of these issues.
Again, here is L. acuarta dosed with over 100ppm K. I apologize that this is not a very good image. Also, this so called "soft water" plant is sitting in a tank with GH 26+, KH 15+, with TDS above 750 microsiemens. Other than the fact that I couldn't get it to turn a rich red color, there were no health problems or structural deformation issues with this plant. The difference in this tank, despite what people think, is simply that the CO2 and flow distribution were Legendary.
T. Barr has publicly challenged me to do the same with R. macandra. It's on my list of answering the challenge of gauntlets thrown, in the manner of the noble Knight, Ivanho.

Cheers,
The carbonate hardness of the water has no bearing on the solubility of gasses in the water, therefore there is no difference in injecting CO2 in the high KH water as opposed to injecting CO2 in low KH water. This is another fallacy that is prevalent throughout the hobby. The solubility of CO2 has nothing to do with alkalinity and nothing to do with carbonate hardness. CO2 solubility in fresh water is strictly limited to the temperature and pressure. Alkalinity and carbonate hardness are not the same thing, and you KH test kit does not measure carbonate hardness, it measure alkalinity, which might be affected by this ion.
If you noticed in Tobi's thread there are those who report increased performance as well as those who report little to no improvement. Again, because many people lack the control as well as lack the reporting skills, it's very difficult to ascertain WHY there is a difference in performance. We need to understand the WHY and that will give us a clue. We do not know for example whether the reporter's tank was previously under-dosed with NO3 and whether the tank was overly lit. Adding urea also produces CO2 as it is converted to NH3->NH4. It could also be that the reporting hobbyist had water that was low in magnesium or that they were using RO without remineralizing and that the simple addition of the Magnesium Nitrate addressed this. NH4 also delivers about 4 times the level of Nitrogen than NO3 does, so of course we should expect an improvement when adding more N per unit mass.
So handing out a sample recipe without any control whatsoever and having people report on results is not really a sound method of collecting data, and drawing a conclusion based on this data is not sound either.
Again, I'm not saying that there is no possibility of excess K+ having a negative effect in certain plants under certain conditions. What I'm saying is that we need better testing to verify the claims and that we should not limit our conclusions to only excess K+ when there are clear examples that fixing the CO2 and distribution, which everyone erroneously assumes is easy, clearly has shown to fix the issue in many cases. Additionally, there are examples of these same plants being grown in high K+ tanks which do not suffer the deformation. Plants have been grown using the ADA system via ADA Aquasoil with the addition of "Brighty K". New ADA aquasoil has about 100X EI levels of NPK, and, Brighty K is a K+ supplement. Viktorlantis, for example is able to grow these plants without any of these issues.
Again, here is L. acuarta dosed with over 100ppm K. I apologize that this is not a very good image. Also, this so called "soft water" plant is sitting in a tank with GH 26+, KH 15+, with TDS above 750 microsiemens. Other than the fact that I couldn't get it to turn a rich red color, there were no health problems or structural deformation issues with this plant. The difference in this tank, despite what people think, is simply that the CO2 and flow distribution were Legendary.
T. Barr has publicly challenged me to do the same with R. macandra. It's on my list of answering the challenge of gauntlets thrown, in the manner of the noble Knight, Ivanho.

Cheers,
NatureBoy
Member
- Joined
- 27 Aug 2008
- Messages
- 374
Hi
Here's my arcuata, with Rotala Macranda narrow in the background

...and from the top

I wish I had taken pics before I had reduced the K+ conc but it looked like it had been struck by a wizards wand! all crumpled and cursed.
Cause and effect is not scientific here I fully agree, but my view isn't based on superstition either. Please bear in mind this is growing in high KH / high TDS tapwater (total hardness 270ppm 15Kh - water quality report).
Instantly this poses a different set of parameters to that of Victor Lantos and ADA who follows the low TDS path as per Tom Barr (all without doubt growing awesome plants!)
(It was my understanding that dissolving carbon dioxide was slightly different to normal dissolution of gases because CO2 forms carbonic acid, and carbonic acid/ bicarbonates / carbonates are governed by equilibrium reactions with Ph determining the relative quantity of each for a given temp, etc. Given Ph is ultimately slightly higher in alkaline water, the injection of CO2 will need to be pushed hard enough to lower the ph before you can safely say that your carbon is in carbonic (utilisable) rather than bicarbonate (non utilisable by certain plants) form on average )
Here's my arcuata, with Rotala Macranda narrow in the background

...and from the top

I wish I had taken pics before I had reduced the K+ conc but it looked like it had been struck by a wizards wand! all crumpled and cursed.
Cause and effect is not scientific here I fully agree, but my view isn't based on superstition either. Please bear in mind this is growing in high KH / high TDS tapwater (total hardness 270ppm 15Kh - water quality report).
Instantly this poses a different set of parameters to that of Victor Lantos and ADA who follows the low TDS path as per Tom Barr (all without doubt growing awesome plants!)
(It was my understanding that dissolving carbon dioxide was slightly different to normal dissolution of gases because CO2 forms carbonic acid, and carbonic acid/ bicarbonates / carbonates are governed by equilibrium reactions with Ph determining the relative quantity of each for a given temp, etc. Given Ph is ultimately slightly higher in alkaline water, the injection of CO2 will need to be pushed hard enough to lower the ph before you can safely say that your carbon is in carbonic (utilisable) rather than bicarbonate (non utilisable by certain plants) form on average )
Hi,
Yes those are beautiful reds that. I was never able to achieve I'm really quite envious of that.
However, the fact that these plants can be grown without issues under both high K+ loading and low K+ loading (whether in the water column or in the sediment) indicates that there is some other factor at play. That's what I'm trying to say. It's not clear in what manner you reduce the K+ loading. Was it simply a withdrawal of KNO3? How long did it take to see the changes, and so forth. How was CO2 distributed? Was there a pruning method that affected the distribution and flow? Was the CO2 reactor in the tank or was it an in-line device? Once the deformation was resolved, was there any attempt to then add a K+ source to see if the deformation returned? I mean, it's possible to reconstruct, but only if we have all the data. It's much easier to disprove a theory that to prove what is. If the plant can be grown successfully in a high K+ environment then that disproves the theory that high K+ loading alone is the cause. This point is super important. Hobbyists suffered all kinds of problems thinking that nutrients caused problems in their tank until someone demonstrated that a problem free tank can be achieved with a high nutrient loading. So we may not be able to say categorically what causes deformation in leaves but we can say what doesn't, and that's just as important.
While it is a fact that CO2 differs from most other gasses in the formation of Carbonic acid, this phenomenon is completely overemphasized, and this leads to misunderstanding. Only a fraction of the gas converts to carbonic acid. Depending on the temperature, somewhere between 1/1000th to 1/600th (0.00001% to 0.000017%) of the gas enters into this carbonic acid / carbonate / bicarbonate equilibrium equation. This is a miniscule amount and is not enough to affect the solubility for our purposes, but is enough to drive a dropchecker for example, because it changes the ratio of protonated hydrogen (H+) ions to hydroxyl (-OH) ions, which is exactly what the pH of the water indicates. There is an entire cult surrounding pH, and most of the cult acolytes don't really know what this number is or how to interpret it, so they do all kinds of crazy things attempting to keep the number constant, never realizing that neither fish, nor plants care.
Furthermore, it is not this miniscule amount of carbonic acid that is used by the plants. The plants do not care about that in this regard. They use the dissolved CO2 which makes up the remaining 99.99999% of the total dissolved gas. In fact, the CO2 must first dissolve into the water in order to drive the carbonic acid equilibrium equation. So this all happens AFTER dissolution and is not an impediment to the gas entering the water.
So people run around in a hysteria about dissolving CO2 in high alkalinity water and they actually do things like purchase RO unit, thinking that soft water dissolves CO2 better than hard water. They change their entire way of life for what amounts to a 0.00001% gain. And, this is why I insist that just because someone thinks that they have fixed their CO2, it does not mean that it has actually been fixed, because there are so many illusions just as powerful as this one.
Cheers,
Yes those are beautiful reds that. I was never able to achieve I'm really quite envious of that.
However, the fact that these plants can be grown without issues under both high K+ loading and low K+ loading (whether in the water column or in the sediment) indicates that there is some other factor at play. That's what I'm trying to say. It's not clear in what manner you reduce the K+ loading. Was it simply a withdrawal of KNO3? How long did it take to see the changes, and so forth. How was CO2 distributed? Was there a pruning method that affected the distribution and flow? Was the CO2 reactor in the tank or was it an in-line device? Once the deformation was resolved, was there any attempt to then add a K+ source to see if the deformation returned? I mean, it's possible to reconstruct, but only if we have all the data. It's much easier to disprove a theory that to prove what is. If the plant can be grown successfully in a high K+ environment then that disproves the theory that high K+ loading alone is the cause. This point is super important. Hobbyists suffered all kinds of problems thinking that nutrients caused problems in their tank until someone demonstrated that a problem free tank can be achieved with a high nutrient loading. So we may not be able to say categorically what causes deformation in leaves but we can say what doesn't, and that's just as important.
While it is a fact that CO2 differs from most other gasses in the formation of Carbonic acid, this phenomenon is completely overemphasized, and this leads to misunderstanding. Only a fraction of the gas converts to carbonic acid. Depending on the temperature, somewhere between 1/1000th to 1/600th (0.00001% to 0.000017%) of the gas enters into this carbonic acid / carbonate / bicarbonate equilibrium equation. This is a miniscule amount and is not enough to affect the solubility for our purposes, but is enough to drive a dropchecker for example, because it changes the ratio of protonated hydrogen (H+) ions to hydroxyl (-OH) ions, which is exactly what the pH of the water indicates. There is an entire cult surrounding pH, and most of the cult acolytes don't really know what this number is or how to interpret it, so they do all kinds of crazy things attempting to keep the number constant, never realizing that neither fish, nor plants care.
Furthermore, it is not this miniscule amount of carbonic acid that is used by the plants. The plants do not care about that in this regard. They use the dissolved CO2 which makes up the remaining 99.99999% of the total dissolved gas. In fact, the CO2 must first dissolve into the water in order to drive the carbonic acid equilibrium equation. So this all happens AFTER dissolution and is not an impediment to the gas entering the water.
So people run around in a hysteria about dissolving CO2 in high alkalinity water and they actually do things like purchase RO unit, thinking that soft water dissolves CO2 better than hard water. They change their entire way of life for what amounts to a 0.00001% gain. And, this is why I insist that just because someone thinks that they have fixed their CO2, it does not mean that it has actually been fixed, because there are so many illusions just as powerful as this one.
Cheers,
NatureBoy
Member
- Joined
- 27 Aug 2008
- Messages
- 374
Hi, Thanks for making my understanding clear on the CO2 / carbonic acid relationship - it was the only part of the process I had not paid that much attention too. I assumed that emphasis was placed on the equilibrium reactions because that was where most CO2 was driven to. As it turns out its rather an academic sideline with regards to CO2 injection! cheers
Regards your questions, I switched from adding KNO3 to daily MgNO3, (I stopped adding MgSO4 as my tapwater is already loaded with sulphates - so essentially not adding anything new, just taking away a large chunk of K, and sulphate) The K I now add is from KH2PO4...in tiny amounts daily (1/8 teaspoon dissolved in 500ml with daily additions of 3ml of this solution to my 270ltr tank). Changes occured within a week, with new growth seemingly "bursting" out of the twisted stunted stems. Importantly I have not seen the deformed growth since making this tweak to fertilising routine...
My substrate is heavy clay topsoil so loaded with minerals plus no doubt a massive amount of bound K+, I've never had to add iron or any trace, ect to get good reds.
CO2 has always been dissolved in line with max mix reactor, nothing over time would suggest that stunting has occured because of CO2 conc related issues (injection rate has been constant - 1hr before lights, off 1hr before lights off - same for when growth was deformed or well formed). On one occasion I felt I could attribute stunting to flow as one stem of Rotala M was seemingly getting smaller when growth was very dense, otherwise I haven't seen a notable correlation between flow and the effect.
Tank is 270ltr volume, with twin 39w T5 osrams from lampspecs. I found Darrel's genius Duckweed Index to be sufficient to monitor any deficiencies etc and do not use test kits.
What is the theory behind K+ effecting Ca 2+ in land based plants, and why would this not be applicable?
cheers
Regards your questions, I switched from adding KNO3 to daily MgNO3, (I stopped adding MgSO4 as my tapwater is already loaded with sulphates - so essentially not adding anything new, just taking away a large chunk of K, and sulphate) The K I now add is from KH2PO4...in tiny amounts daily (1/8 teaspoon dissolved in 500ml with daily additions of 3ml of this solution to my 270ltr tank). Changes occured within a week, with new growth seemingly "bursting" out of the twisted stunted stems. Importantly I have not seen the deformed growth since making this tweak to fertilising routine...
My substrate is heavy clay topsoil so loaded with minerals plus no doubt a massive amount of bound K+, I've never had to add iron or any trace, ect to get good reds.
CO2 has always been dissolved in line with max mix reactor, nothing over time would suggest that stunting has occured because of CO2 conc related issues (injection rate has been constant - 1hr before lights, off 1hr before lights off - same for when growth was deformed or well formed). On one occasion I felt I could attribute stunting to flow as one stem of Rotala M was seemingly getting smaller when growth was very dense, otherwise I haven't seen a notable correlation between flow and the effect.
Tank is 270ltr volume, with twin 39w T5 osrams from lampspecs. I found Darrel's genius Duckweed Index to be sufficient to monitor any deficiencies etc and do not use test kits.
What is the theory behind K+ effecting Ca 2+ in land based plants, and why would this not be applicable?
cheers
Hi,
Well, the activity of Calcium is not all that well understood. There are two fundamental differences between these two ions that may give a clue, and this is what is so bizarre about the idea of K+ toxicity:
High Ca++ levels in the cytoplasm of the plant is extremely toxic.
The plant develops some very restrictive Ca++ uptake mechanism to counter the threat.
Ca++ is very mobile within the leaf but is does not travel from the leaf. Over the life of the leaf Ca++ does not leave but only accumulates.
Potassium concentrations with the plant is extremely high because K+ is not toxic and is used in almost every function of the plant. Since it is not toxic the plant uses the buildup of K+ to act as a "battery electrolyte". The electrochemical charge of local high K+ concentration provides the electrical potential to move negatively charge ions across cell membranes. So K+ is ubiquitous.
K+ is extremely mobile within the plant and is found everywhere in extremely high concentrations
There are many different channels (or pathways) for K+ uptake.
In terrestrial plants the roots create a voltage potential at the channel site in order to attract the ions. If Ca++ concentration in the soil is low but K+ concentration levels are high then K+ overwhelms the uptake mechanism and gets picked up faster than Ca++.
It could possibly be therefore that since only soil uptake is possible for land plants the combination of high K+ mobility/activity when combined with the more restrictive Ca++ uptake mechanism, reduce the ability of the plant to properly uptake Ca++. In aquatic plants the Ca++ can enter the leaf directly from the water as well as from the substrate so there does not seem to be the "bottleneck potential" which is present when only sediment uptake is available. Again, this is all theoretical.
In any case, there is so much Potassium in plants that when you burn them, the remaining ashes are always very high in K++. Hundreds of years ago, wood and other organic matter was burned in pots in the process of fabricating soap. This burnt residue, or, ash was called unsurprisingly, "Pot Ashes" and was basically a cocktail of various Potassium salts. After rinsing, the ashes were dried and later combined with animal fat to produce the soap. Pot ashes, later became known as Potash and is therefore associated with K ---- in effect Pot_ash_ium or Potassium. Now, any salt of K is referred to as a type of Potash, so for example the expression "Potash of Nitrate" is simply Potassium Nitrate (KNO3) and Potash of Sulfate is Potassium Sulfate (K2SO4).
Because plants contain such high concentration of potassium, and since it is used in so many locations and so many functions, it is difficult to accept the argument that high K+ concentration in the water column is toxic, but blocking of other ions is a generally accepted idea, on land. Whether it is actually valid I really don't know. It may be true for some species but definitely not for others.
Cheers,
Well, the activity of Calcium is not all that well understood. There are two fundamental differences between these two ions that may give a clue, and this is what is so bizarre about the idea of K+ toxicity:
High Ca++ levels in the cytoplasm of the plant is extremely toxic.
The plant develops some very restrictive Ca++ uptake mechanism to counter the threat.
Ca++ is very mobile within the leaf but is does not travel from the leaf. Over the life of the leaf Ca++ does not leave but only accumulates.
Potassium concentrations with the plant is extremely high because K+ is not toxic and is used in almost every function of the plant. Since it is not toxic the plant uses the buildup of K+ to act as a "battery electrolyte". The electrochemical charge of local high K+ concentration provides the electrical potential to move negatively charge ions across cell membranes. So K+ is ubiquitous.
K+ is extremely mobile within the plant and is found everywhere in extremely high concentrations
There are many different channels (or pathways) for K+ uptake.
In terrestrial plants the roots create a voltage potential at the channel site in order to attract the ions. If Ca++ concentration in the soil is low but K+ concentration levels are high then K+ overwhelms the uptake mechanism and gets picked up faster than Ca++.
It could possibly be therefore that since only soil uptake is possible for land plants the combination of high K+ mobility/activity when combined with the more restrictive Ca++ uptake mechanism, reduce the ability of the plant to properly uptake Ca++. In aquatic plants the Ca++ can enter the leaf directly from the water as well as from the substrate so there does not seem to be the "bottleneck potential" which is present when only sediment uptake is available. Again, this is all theoretical.
In any case, there is so much Potassium in plants that when you burn them, the remaining ashes are always very high in K++. Hundreds of years ago, wood and other organic matter was burned in pots in the process of fabricating soap. This burnt residue, or, ash was called unsurprisingly, "Pot Ashes" and was basically a cocktail of various Potassium salts. After rinsing, the ashes were dried and later combined with animal fat to produce the soap. Pot ashes, later became known as Potash and is therefore associated with K ---- in effect Pot_ash_ium or Potassium. Now, any salt of K is referred to as a type of Potash, so for example the expression "Potash of Nitrate" is simply Potassium Nitrate (KNO3) and Potash of Sulfate is Potassium Sulfate (K2SO4).
Because plants contain such high concentration of potassium, and since it is used in so many locations and so many functions, it is difficult to accept the argument that high K+ concentration in the water column is toxic, but blocking of other ions is a generally accepted idea, on land. Whether it is actually valid I really don't know. It may be true for some species but definitely not for others.
Cheers,
NatureBoy
Member
- Joined
- 27 Aug 2008
- Messages
- 374
There are many different channels (or pathways) for K+ uptake.
In terrestrial plants the roots create a voltage potential at the channel site in order to attract the ions. If Ca++ concentration in the soil is low but K+ concentration levels are high then K+ overwhelms the uptake mechanism and gets picked up faster than Ca++.
It could possibly be therefore that since only soil uptake is possible for land plants the combination of high K+ mobility/activity when combined with the more restrictive Ca++ uptake mechanism, reduce the ability of the plant to properly uptake Ca++. In aquatic plants the Ca++ can enter the leaf directly from the water as well as from the substrate so there does not seem to be the "bottleneck potential" which is present when only sediment uptake is available. Again, this is all theoretical.
Many thanks for your post.
Through observing these aquatic stem plants and the way they grow it's occurred to me that there could well be some calcium supply system happening through the xylem in a similar way to terrestrial plants to the growing point of aquatic plants (rather than calcium uptake from the water column directly at the growing point)...otherwise how does the stem plant manage to produce emergent growth, something that L arcuata is particularly fond of doing, even from a submerged stem?
Calcium needs to be pumped up through the stem to the emergent growing point. It's conceivable that this same mechanism could supply the calcium for the formation of all leaves too - including submerged leaves. If so, then it opens the door for the same type of K+, Ca ++, ion competitive uptake issues that have been reported for terrestrial plants where a higher concentration of potassium inhibits the uptake of calcium.
cheers
plantbrain
Member
- Joined
- 2 Aug 2007
- Messages
- 1,938
I agree that the vast majority of aquatic plants flourish without any adverse regulatory issues regarding K+ concentrations in the water column, certainly true of the Pogostemon I've had.
However certain plants, perhaps those adapted to water with extremely low "hardwater" ion concentrations may not be able to regulate the K+ ion so efficiently. Plants that come to mind where I've seen this most apparent are L arcuata, R macaranda - when I lay off the K+, normal leaf formation, all other factors constant (as possible😉)
I don't suggest this simply to be contentious, but observing growth under different concentrations of nutrients / lights / CO2 has led me to this opinion.
I'm not a potassium "hater", rather a potassium respecter.
Alright, let's have some names of these plants?
P. stellata comes from rain fed low conductivity waters, many plants do, this does not imply they have any trouble with high K+ in the water column, it simply implied there's not much available in the water column.
There may be plenty in the soil. Or it maybe a flowing system where 0.01 ppm K+ is constantly being replaced. Natural systems demonstrate nothing as far as preference or optimal growing conditions. All it shows is that is where that plant can exist at that point in time and within that space.
If you want to ask the question or pose a hypothesis, I'll indulge:
At what concentration will we see K+ stunting or algae or a reduction in growth due to K+ independent of other variables?
I need numbers, and I need names of the plants.
If you cannot produce it, it's not a question of hater/respecter, belief................... it's a question of basic common sense and logic.
Now if you can do the work and show out of some 400 species I think I'm up to at the present time, not one species has shown any evidence of issues at higher concentrations for BOTH locations(soil or the water column), I think the burder of proof and naming the ppm's and the species in question that will noit like or show less than optimal growing conditions at those ppm's. I have never been proven wrong on this yet. I might, but no one has even come remotely close in some 2 decades.
I make no assumptions about plants. I do have the experience however and KNOW, beyond any reasonable doubt, that I've added some rich K+, not as crazy as Clive mind you, but at 50-100ppm ranges, that's a pretty massive target to hit.
If folks cannot target 10-50ppm of K+, then they likely are in the wrong hobby.
the principle of falsification does not say what causes your or my stunting, it only states what it cannot possibly be.
So it's rather a simple matter to test if a plant is sensitive to various K+ levels.
Deducing what someone else's issues in their tank when they dose something is quite another matter, they dose something and see dependencies. That suggest they have other issues and do not have a reference control. Most hobbyists tend to be in that boat, not having mastered good optimal growth.
Falsification does not answer what their issues are/maybe, it only removes things like K+, or PO4 excess etc. All it takes are few folks that dose to say 50-100ppm and have the plants looking nice/sell them often to others etc.
Then you toss that hypothesis out the window and look for some other cause/make a new hypothesis to test. If you are in a local club and you know someone like Clive, you see his tanks in person and see him dose, then there's no belief about it.
You know that cannot be a valid hypothesis.
I dose about 35-40 ppm a week to my tanks as K+,m but I do larger water changes, 70-80%.
So there is much less build up beyond that. I doubt many need more than say 25 ppm a week.
A simple dosing calculator can easily help anyone target that amount.
In general, you'd have to have 75% of your N coming from the sediment or fish waste etc, before you'd run low on K+ if you use KNO3 as a source of NO3.
I do not add K2SO4 etc extra etc, some folks do, but they have no issues. Folks use to dose K+ like mad men in 1990's till about 2002 or so. Then myths started being sent around the web.
There's quite a few things that could be going on with the crowd that believes K+ inhibits things:
Enzyme machinery inside plants needs to adapt and if you bob at lower levels, you can limit the plant oscillating between two levels.
If you supply plenty of K+ at a high level, then there's no issue.
Liebig's law always applies. Many aquarist seem okay with complete denial that this law exist. If the CO2 was just a little limiting when you had high ppm's of K+, say 30 ppm, and you lower P or K, then you might be tempted to assuem that it's plants liking lower K+ ppm, and not even think about Liebig or CO2.
When another hobbyists comes along and dose the same thing and reports no such issues, then it cannot be what the 1st guy assumed. He had confounding errors.
So there's 2 things that are overlooked by folks claiming this, but even still, you cannot argue the fact that folks dose plenty of K+ without any issues, this claim cannot be true.
Those are the facts, see what conclusions you can draw from them.
You do not go here are my conclusions, let's see what facts we can find to support them.
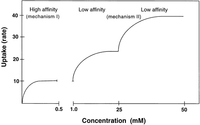
plantbrain
Member
- Joined
- 2 Aug 2007
- Messages
- 1,938
I switched from adding KNO3 to daily MgNO3, (I stopped adding MgSO4 as my tapwater is already loaded with sulphates - so essentially not adding anything new, just taking away a large chunk of K, and sulphate) The K I now add is from KH2PO4...in tiny amounts daily (1/8 teaspoon dissolved in 500ml with daily additions of 3ml of this solution to my 270ltr tank). Changes occured within a week, with new growth seemingly "bursting" out of the twisted stunted stems. Importantly I have not seen the deformed growth since making this tweak to fertilising routine...
What is the theory behind K+ effecting Ca 2+ in land based plants, and why would this not be applicable?
cheers
So this (Toby's claim and special N) is the latest source of the K+ myth?
History repeated yet again.
About every 2-5 years, a cohort repeats the same old myths over and over.
Liebig's law applies.
If you limit one nutrient, and see a response...........it often takes the demand/stress off another, mostly CO2.
I've grow Rotala macrandra🙂

The species below make macrandra look like beginner plants:

30-40ppm of K+.
Not found a single species yet that is touchy to K+ over this range.
Or high NO3, or high Fe, or high PO4, Ca, Mg.
KH?
CO2?
Yes!!!!!!
I cannot rule those out.
NatureBoy
Member
- Joined
- 27 Aug 2008
- Messages
- 374
Hi Tom
Many thanks for your input. To you and Clive, this conversation is madness, as you've proven to yourself that all said plants grow well with your tank parameters with + 30-40ppm K+. I can confirm that pogostemon grows crazy the more K+, phosphate you can throw at it, same with the majority of plants.
My thoughts on K have been based on my own experiences with it, particularly with Ludwigia Arcuata and R Macranda, when I've looked around I've seen others having issues with it too, this is over the last ten years or so. Over that time I've added higher K and when I do, some plants just don't seem to like it.
Perhaps in accordance with Liebig's law by adding more potassium I've unlocked a limiting factor / created luxury uptake and as the rate of growth cannot continue to be supported with the current level of CO2, then I see the growth go deformed, a bit like turning the sausage machine on faster than the guy can make the sausage. Who's at fault?
Different sets of parameters lead to different conclusions it seems. I'm growing rotala macranda in tapwater with KH15, and it grows fast. In the spirit of falsification I've been inclined to dismiss KH as a concern...so long as I keep the K+ down.... !
!
Perhaps what this amounts to is I'm happy, with a given level of CO2, to have potassium as a limiting factor on growth, given I provide enough for basic function.
cheers all
Many thanks for your input. To you and Clive, this conversation is madness, as you've proven to yourself that all said plants grow well with your tank parameters with + 30-40ppm K+. I can confirm that pogostemon grows crazy the more K+, phosphate you can throw at it, same with the majority of plants.
My thoughts on K have been based on my own experiences with it, particularly with Ludwigia Arcuata and R Macranda, when I've looked around I've seen others having issues with it too, this is over the last ten years or so. Over that time I've added higher K and when I do, some plants just don't seem to like it.
Perhaps in accordance with Liebig's law by adding more potassium I've unlocked a limiting factor / created luxury uptake and as the rate of growth cannot continue to be supported with the current level of CO2, then I see the growth go deformed, a bit like turning the sausage machine on faster than the guy can make the sausage. Who's at fault?
Different sets of parameters lead to different conclusions it seems. I'm growing rotala macranda in tapwater with KH15, and it grows fast. In the spirit of falsification I've been inclined to dismiss KH as a concern...so long as I keep the K+ down....
 !
!Perhaps what this amounts to is I'm happy, with a given level of CO2, to have potassium as a limiting factor on growth, given I provide enough for basic function.
cheers all


