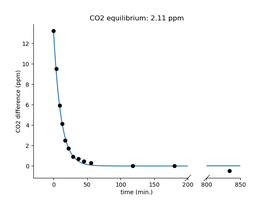
Last week, I ran an experiment to see what the CO2 concentration will be in water left to equilibrate on air and how quickly we can reach the equilibrium with or without agitation. The experiment was a bit sloppy, so I plan to repeat it, but this time with a little more careful planning of the conditions and splitting different questions into different experiments. Also, I have a few ideas and questions that can be tested with a single pH meter so I will collect those experiments here. Any contribution, critics, ideas, or measurement repeats are welcome. I am unsure if answering the questions below would result in practical benefits, but a better understanding of the aquarium systems can not hurt. Anyway, I am curious if anything comes out of these. So, here are the initial ideas/questions that I think could be tested, not all related to each other:
1. Equilibrium CO2 concentration in the water left in the air. I already tested this, but I don't know if I have waited enough, and also, I did not calculate with the evaporated volume. We could also simply calculate it using Henry's law, especially if we know the CO2 levels in our room, but still, I think it is worth seeing it for ourselves. The equilibrium level is also required for some of the further experiments.
2. Testing the dynamics of the CO2 exchange. How quickly does the equilibrium happen? How will the water's volume, surface area, and agitation influence this? This is the second question I tried to approximate last time, but I would separate this question from the equilibrium measurement. I'd like to test whether we can use Fick's law to calculate/predict the CO2 exchange rate between water and air. If so, can we come up with a simple test to describe the gas exchange rate (actually the diffusion capacity) in any specific aquarium?
3. Can we estimate the CO2 production in a specific tank using pH and KH measurements only? If we know the diffusion capacity of an aquarium, and the actual CO2 levels in it (and in the air), then this should be doable.
4. Can we estimate the average O2 levels in an aquarium based on the CO2 production and the gas exchange rate in an aquarium?
5. Can we estimate the level of non-bicarbonate buffers in our tank? Non-bicarbonate buffers (phosphate and organic compounds) will modify the readings of our KH tests and result in false CO2 calculations.
6. Does the CO2 have better solubility/higher levels in water with low KH than in one with high KH? I don't think so, but this idea has been circulating for a while, and it is easy to test. If the equilibrium CO2 levels are not different, is the diffusion rate of CO2 different? There is probably already data for that somewhere, but still, I would like to test that too.
The idea of some of these tests comes from identical testing in human medicine. In human blood, similarly to aquariums and natural aquatic systems, the main buffer is the bicarbonate, along with some phosphate and organic molecules (proteins). The pH is regulated through lung ventilation (affecting the CO2 levels in the blood). The modern human respiratory acid-base physiology was born in 1950s during the poliomyelitis pandemic with the need to determine the CO2 levels in the blood to estimate the ventilation need of the patients. It turned out that one can do that by measuring pH only... Based on pH measurements, CO2 levels, bicarbonate concentration, and the concentration of other non-bicarbonate buffer levels can be determined using the basic principles and equations of the CO2/pH/bicarbonate connection. The diffusion capacity is also determined nowadays for patients with lung diseases to estimate the capacity of the lungs to exchange CO2 for O2. In aquariums, it could describe how well the aquarium is aerated with a number that could be comparable between aquariums. We need some methodological modifications for aquarium water testing at home, but maybe we can come up with a strategy to get estimations for the above questions.
Equilibrium CO2 levels in water.
The first experiment would be a reproduction from the last week, with some modifications:
1. The aim is to measure the equilibrium CO2 levels; no dynamic changes
2. To avoid water heating, the water will be aerated with an air pump instead of a small pump. I will use a calculator which considers the temperature.
3. Two water samples will be used, both aerated and having 10 dKH hardness set with NaCO3. In one of them I will use water as it comes out of the RO device, while the other will be boiled for an hour to degas it. Hopefully, one will reach the equilibrium CO2 concentration from above, the other from below. The pH in the two jars should be equal at the equilibrium concentrations.
4. I plan to measure the jars with water before the start and the end to get the amount of evaporated water. Then, the bicarbonate concentration can be corrected.
Any recommendations?
1. Equilibrium CO2 concentration in the water left in the air. I already tested this, but I don't know if I have waited enough, and also, I did not calculate with the evaporated volume. We could also simply calculate it using Henry's law, especially if we know the CO2 levels in our room, but still, I think it is worth seeing it for ourselves. The equilibrium level is also required for some of the further experiments.
2. Testing the dynamics of the CO2 exchange. How quickly does the equilibrium happen? How will the water's volume, surface area, and agitation influence this? This is the second question I tried to approximate last time, but I would separate this question from the equilibrium measurement. I'd like to test whether we can use Fick's law to calculate/predict the CO2 exchange rate between water and air. If so, can we come up with a simple test to describe the gas exchange rate (actually the diffusion capacity) in any specific aquarium?
3. Can we estimate the CO2 production in a specific tank using pH and KH measurements only? If we know the diffusion capacity of an aquarium, and the actual CO2 levels in it (and in the air), then this should be doable.
4. Can we estimate the average O2 levels in an aquarium based on the CO2 production and the gas exchange rate in an aquarium?
5. Can we estimate the level of non-bicarbonate buffers in our tank? Non-bicarbonate buffers (phosphate and organic compounds) will modify the readings of our KH tests and result in false CO2 calculations.
6. Does the CO2 have better solubility/higher levels in water with low KH than in one with high KH? I don't think so, but this idea has been circulating for a while, and it is easy to test. If the equilibrium CO2 levels are not different, is the diffusion rate of CO2 different? There is probably already data for that somewhere, but still, I would like to test that too.
The idea of some of these tests comes from identical testing in human medicine. In human blood, similarly to aquariums and natural aquatic systems, the main buffer is the bicarbonate, along with some phosphate and organic molecules (proteins). The pH is regulated through lung ventilation (affecting the CO2 levels in the blood). The modern human respiratory acid-base physiology was born in 1950s during the poliomyelitis pandemic with the need to determine the CO2 levels in the blood to estimate the ventilation need of the patients. It turned out that one can do that by measuring pH only... Based on pH measurements, CO2 levels, bicarbonate concentration, and the concentration of other non-bicarbonate buffer levels can be determined using the basic principles and equations of the CO2/pH/bicarbonate connection. The diffusion capacity is also determined nowadays for patients with lung diseases to estimate the capacity of the lungs to exchange CO2 for O2. In aquariums, it could describe how well the aquarium is aerated with a number that could be comparable between aquariums. We need some methodological modifications for aquarium water testing at home, but maybe we can come up with a strategy to get estimations for the above questions.
Equilibrium CO2 levels in water.
The first experiment would be a reproduction from the last week, with some modifications:
1. The aim is to measure the equilibrium CO2 levels; no dynamic changes
2. To avoid water heating, the water will be aerated with an air pump instead of a small pump. I will use a calculator which considers the temperature.
3. Two water samples will be used, both aerated and having 10 dKH hardness set with NaCO3. In one of them I will use water as it comes out of the RO device, while the other will be boiled for an hour to degas it. Hopefully, one will reach the equilibrium CO2 concentration from above, the other from below. The pH in the two jars should be equal at the equilibrium concentrations.
4. I plan to measure the jars with water before the start and the end to get the amount of evaporated water. Then, the bicarbonate concentration can be corrected.
Any recommendations?