Hi friends, the sale of ammonium nitrate is prohibited in my country. I have ammonium sulfate and magnesium nitrate, potassium nitrate. If I want to make original 33% ammonium nitrate, what kind of mixture should I make?I don't know the exact percentage of ammonium nitrate.
-
You are viewing the forum as a Guest, please login (you can use your Facebook, Twitter, Google or Microsoft account to login) or register using this link: Log in or Sign Up
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
make ammonium nitrate
- Thread starter hasan66
- Start date
Hi all,
Can you get <"urea (CO(NH2)2)">? Personally I would be much happier <"using urea as a TAN source">, purely in fish welfare terms.
cheers Darrel
That is the <"same in a lot of countries">, all nitrate compounds are potential explosives, but some give you a bigger bang.the sale of ammonium nitrate is prohibited in my country.
Can you get <"urea (CO(NH2)2)">? Personally I would be much happier <"using urea as a TAN source">, purely in fish welfare terms.
You need the RMM of NH4NO3 = (14 + 4) + (14 + 48) = 80. The NH4 is 18 / 80 = 22.5% and NO3 bit is 62 / 80 = 77.5%. The nitrogen content is 28 / 80 = 35%.If I want to make original 33% ammonium nitrate, what kind of mixture should I make?
You can work out the percentages of any other compounds in <"exactly the same way">.I have ammonium sulfate and magnesium nitrate, potassium nitrate
cheers Darrel
MichaelJ
Member
all nitrate compounds are potential explosives, but some give you a bigger bang.
By itself Ammonium Nitrate (and other common nitrates) is not an explosive, but an oxidizer - that is, it greatly accelerates combustion when mixed with an explosive or flammable compound - NH4NO3 being a particular powerful oxidizer.
Personally, I would go with what Darrel suggest above and use Urea or use a fertilizer that contains NH4NO3 such as Tropica Specialized.
Keep in mind that even the mere production of Ammonium Nitrate in private may be prohibited in your country!
Cheers,
Michael
Last edited:
John q
Member
Yep, don't store ( old) ammonium nitrate next to "dodgy" munitions 😃That was a pretty big bang in Beirut!
I think the op is well versed in dosing Urea and calcium ammonium nitrate, so not 100% sure why they are chasing a ammonium nitrate "recipe" 🥺
Alright friend, I will continue on my way with urea.Hi all,
That is the <"same in a lot of countries">, all nitrate compounds are potential explosives, but some give you a bigger bang.
Can you get <"urea (CO(NH2)2)">? Personally I would be much happier <"using urea as a TAN source">, purely in fish welfare terms.
You need the RMM of NH4NO3 = (14 + 4) + (14 + 48) = 80. The NH4 is 18 / 80 = 22.5% and NO3 bit is 62 / 80 = 77.5%. The nitrogen content is 28 / 80 = 35%.
You can work out the percentages of any other compounds in <"exactly the same way">.
cheers Darrel
My friend, there is 1 percent ammonium in the fertilizer. My nitrate-based rotalas do not like nitrate. When I give too much nitrate, I experience distortion in the top parts. It shrinks and becomes stunted. I attribute this to the excessive dose of No3.Yep, don't store ( old) ammonium nitrate next to "dodgy" munitions 😃
I think the op is well versed in dosing Urea and calcium ammonium nitrate, so not 100% sure why they are chasing a ammonium nitrate "recipe" 🥺
John q
Member
Well urea is approx 46% N, do the nitrate intolerant rotala not like this source? CAN contains about 27% N, half of which comes from ammonium, so not sure where the 1% figure you mention comes from.
am not gonna lie but NH4NO3 is one of the best Nitrogen you could use for aquatic plant, even the NH4Cl or (NH4)2SO4 cannot replace it. But, Urea isn't too far behind and I would use Urea as my source of Nitrogen when NH4NO3 isn't available. it is also very safe compare to directly adding the NH4 when used properly.
mostly common in those tanks where they lack Ammonium or Ammonium based soil, especially when the main source of Nitrogen is NO3.My friend, there is 1 percent ammonium in the fertilizer. My nitrate-based rotalas do not like nitrate. When I give too much nitrate, I experience distortion in the top parts. It shrinks and becomes stunted. I attribute this to the excessive dose of No3.
MichaelJ
Member
Hi @Happi, That is my understanding as well... NH4NO3 is basically one of the most easily absorbable sources of Nitrogen we can provide for our aquatic plants (2 Nitrogen atoms only bonded by 4 simple Hydrogen atoms and 3 Oxygen atoms) … and the plants don't need much.am not gonna lie but NH4NO3 is one of the best Nitrogen you could use for aquatic plant, even the NH4Cl or (NH4)2SO4 cannot replace it. But, Urea isn't too far behind and I would use Urea as my source of Nitrogen when NH4NO3 isn't available. it is also very safe compare to directly adding the NH4 when used properly.
Cheers,
Michael
Last edited:
Yes, my soil is not ammonium based, so when I give the NO3 dose, the plants are unhappy, but when I give the NH4 dose, everyone is happy, but they are happy only up to a certain point. That's why I thought I should make a half ammonium half nitrate source that they will love, so I use urea fertilizer in addition to calcium ammonium nitrate. I will add it and try it like thismostly common in those tanks where they lack Ammonium or Ammonium based soil, especially when the main source of Nitrogen is NO3.
Peki üre yaklaşık %46 N'dir, nitrat intoleransı olan rotala bu kaynağı sevmiyor mu? CAN, yarısı amonyumdan gelen yaklaşık %27 N içerir, dolayısıyla bahsettiğiniz %1 rakamının nereden geldiğinden emin değilim.
Attachments
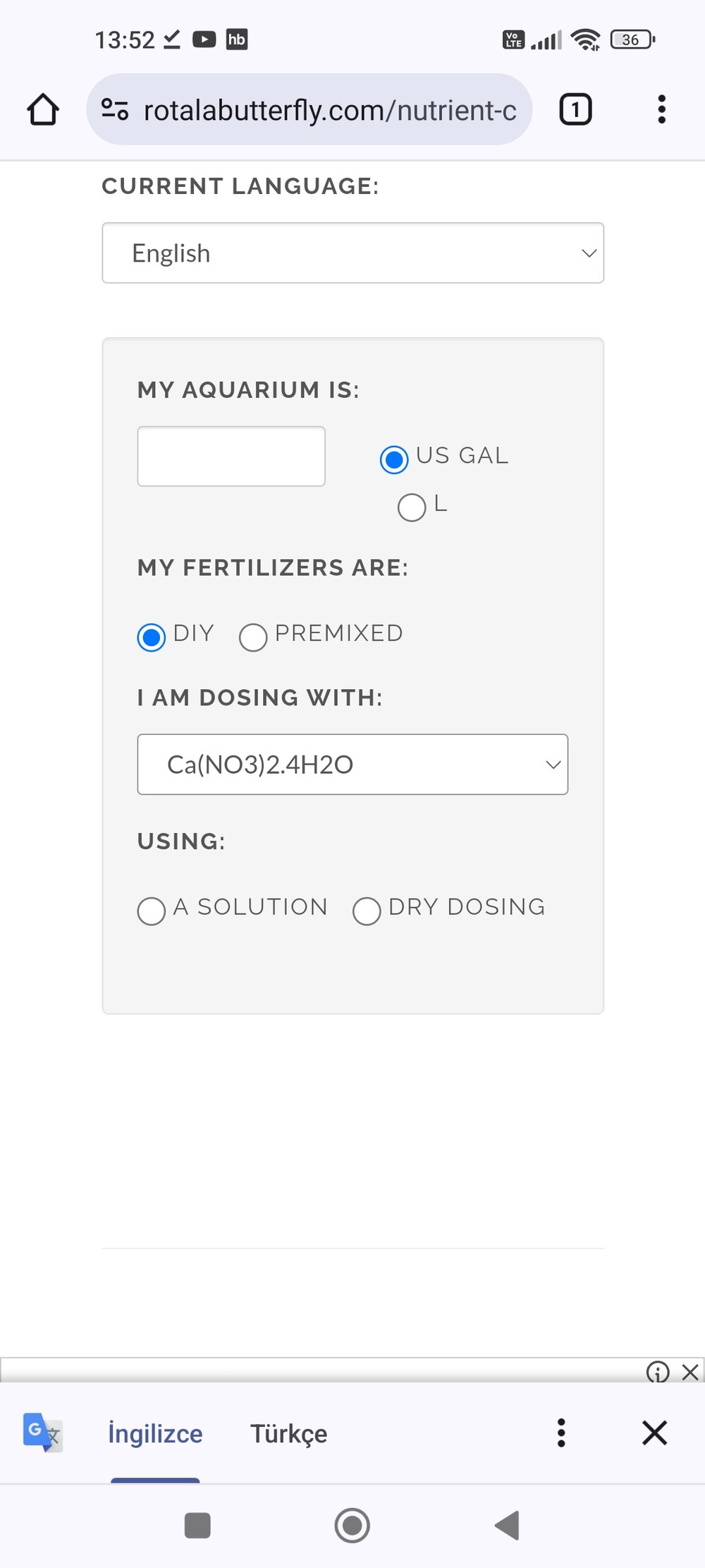
I'm laughing at the mistake I made right nowIt appears what you have in the picture above @hasan66 is Calcium Nitrate, not Calcium ammonium nitrate. That would indeed explain the 1%.
Would seem we are at cross purposes here.
But it contains ammonia. Is this compound found in the 1% calculator?
But it contains ammonia. Is this compound found in the 1% calculator?It appears what you have in the picture above @hasan66 is Calcium Nitrate, not Calcium ammonium nitrate. That would indeed explain the 1%.
Would seem we are at cross purposes here.
Hi all,
This means that you can't really use the calculator as such, you would need to convert the result you get by the difference between the nitrogen content of your fertiliser and the salt you use in the <"Rotala Nutrient Calculator">.
Having said that the result from "calcium nitrate tetrahydrate" Ca(NO3)2.4H2O will be pretty close.
RAM
Ca = 40.1, N = 14, O = 16, H = 1
RMM
40.1 + (14*2) 28 + (16 * 6) 96 + (8) + (4 * 16)64 = 236.1 and 40.1 / 236.1 = 17% N and 17 / 15.5 = ~ 1.1 so 10g of Ca(NO3)2.4H2O has the same amount of nitrogen as ~ 11g "NitroChalk".
cheers Darrel
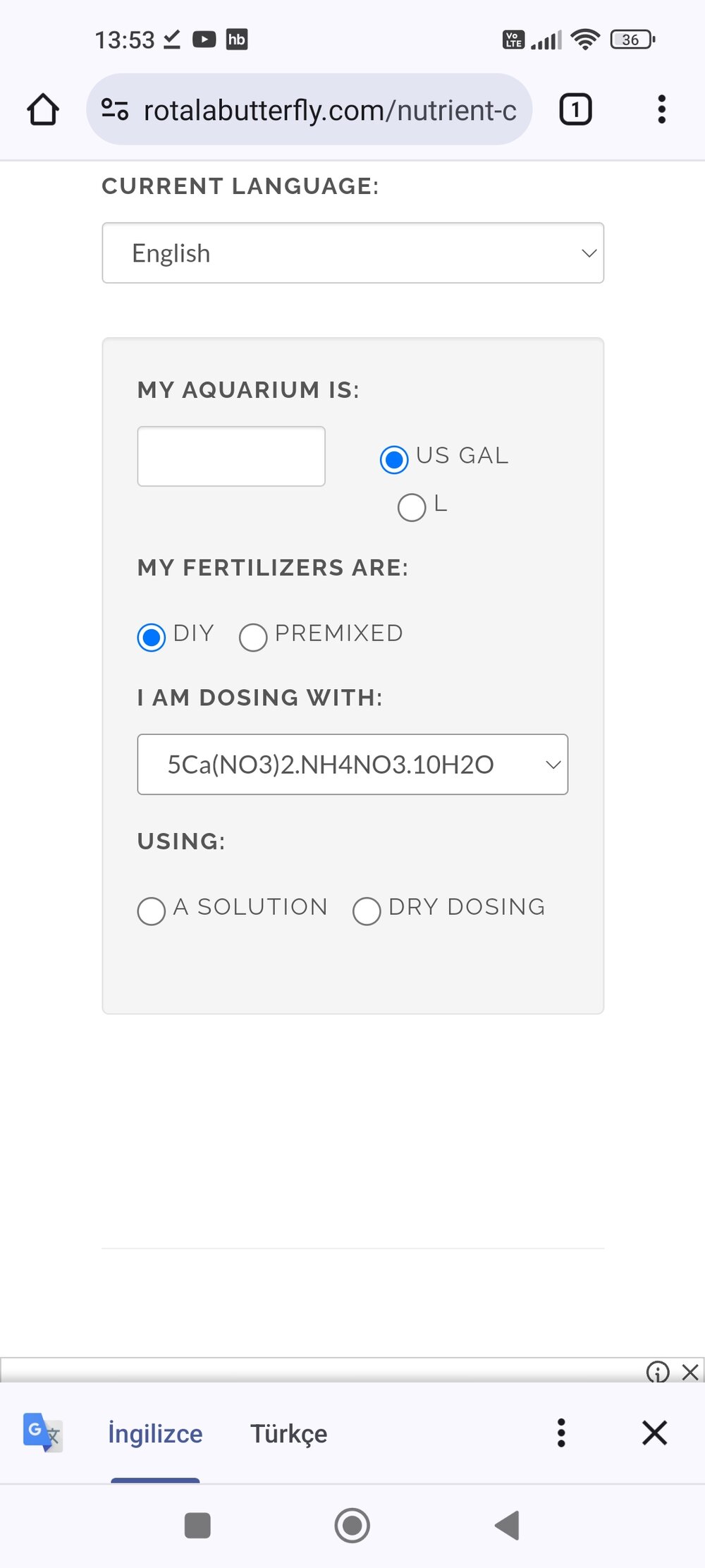
Unfortunately <"it is a bit complicated">, but because your fertiliser is 15 or 16% nitrogen (N), which means you have <"NitroChalk">, which is probably the "hydrated double salt - 5Ca(NO3)2•NH4NO3•10H2O" which still doesn't quite work, some I'm going to guess plus some unreacted CaCO3.But it contains ammonia. Is this compound found in the 1% calculator?
This means that you can't really use the calculator as such, you would need to convert the result you get by the difference between the nitrogen content of your fertiliser and the salt you use in the <"Rotala Nutrient Calculator">.
Having said that the result from "calcium nitrate tetrahydrate" Ca(NO3)2.4H2O will be pretty close.
RAM
Ca = 40.1, N = 14, O = 16, H = 1
RMM
40.1 + (14*2) 28 + (16 * 6) 96 + (8) + (4 * 16)64 = 236.1 and 40.1 / 236.1 = 17% N and 17 / 15.5 = ~ 1.1 so 10g of Ca(NO3)2.4H2O has the same amount of nitrogen as ~ 11g "NitroChalk".
I think, if you don't have any livestock, ammonium nitrate (NH4NO3) would be the best option for the reasons that @MichaelJ mentions. It is commercial growers "nitrogen source of choice" and they are a <"pretty good indication">.am not gonna lie but NH4NO3 is one of the best Nitrogen you could use for aquatic plant
There are inherent limitations of <"commercial fertiliser mixes">, but there is also the disclaimer that commercial hydroponic fertilisers must fulfill the basic requirement of promoting plant growth, or they companies that make them would have gone out of business.
Personally I wouldn't use the "Nitrochalk" , I'd <"use urea"> (CO(NH2)2) and <"another calcium source">.But, Urea isn't too far behind and I would use Urea as my source of Nitrogen when NH4NO3 isn't available. it is also very safe compare to directly adding the NH4 when used properly.
cheers Darrel
I told the seller that I wanted calcium nitrate and he gave me this and I have one 100 liter tank and one 75 liter tank. I don't know how many grams to use in my 500ml container. I want to get 1ml 1ppm. I am faced with a very complicated situation. I am bad at mathematics. I have a hard time understanding your calculations.