-
You are viewing the forum as a Guest, please login (you can use your Facebook, Twitter, Google or Microsoft account to login) or register using this link: Log in or Sign Up
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Ph drop & high alkalinity
- Thread starter RossMartin
- Start date
Hi all,
The hardness of the water (or its pH) shouldn't make any difference, as long as you have 4dKH indicator solution in your drop checker.
When you have 4dKH solution + bromothymol blue indicator, you can use the experimentally defined equilibrium values between dKH ("bicarbonate" HCO3-) and CO2 (as "carbonic acid" H2CO3), to give you the level of CO2 in the tank, (nb. only a very small proportion of CO2 becomes H2CO3, 99.8% remains as the dissolved gas).
We use bromothymol blue because it is a narrow range pH indicator, yellow at pH6, green at pH7 and blue at pH8. Table below.
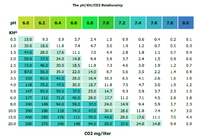
Why we need to use 4dKH indicator solution in the drop checker
This is really important, if you had tank water with 20dKH it would naturally be at ~ pH7.8 (at the equilibrium value between HCO3- and atmospheric CO2 levels), but it is almost endlessly carbonate buffered, and if we then added enough CO2 to depress the pH to pH6.8 we would have 94ppm of CO2 and a lot of dead fish.
The Bouncy Castle analogy
If that doesn't make sense and I don't know if this helps, but I'll use the "Bouncy Castle" analogy where "compressed air" substitutes for "added CO2".
When we aren't actively pumping air into the bouncy castle via a compressor, it is deflated and the air pressure inside and outside are the same, this is analogous to a fish tank where we aren't adding CO2.
When we turn the compressor on air is pumped into the castle and it inflates, in the case of CO2, when we add CO2 the HCO3- ~ H2CO3 equilibrium is driven towards H2CO3, you now have an extra H+ ion, and acids are defined as H+ donors, so the pH falls (the water becomes more acid).
If our bouncy castle doesn't have any holes in it, we don't need to add much compressed air to keep it inflated, or in the CO2 case this is the low dKH scenario where a small amount of CO2 addition causes a large pH drop. If the castle has a lot of holes we need to add a lot air to keep it inflated, and in the CO2 case this is the high dKH scenario.
But, rather than estimating how many holes we have, we can have a hole of standard size that always lets out the same amount of air, for CO2 this is the "4dKH solution in the drop checker scenario", where we add a stable amount of air ("CO2") to keep the castle inflated ("CO2 at 25ppm").
In all these cases if we turn the compressed air (CO2) off, the castle deflates and the HCO3- ~ H2CO3 equilibrium returns to its atmospheric level.
Personally for me the air compressor is always going to be turned off, as I can find quite enough ways to accidentally kill my fish without deliberately adding another one.
cheers Darrel
The hardness of the water (or its pH) shouldn't make any difference, as long as you have 4dKH indicator solution in your drop checker.
When you have 4dKH solution + bromothymol blue indicator, you can use the experimentally defined equilibrium values between dKH ("bicarbonate" HCO3-) and CO2 (as "carbonic acid" H2CO3), to give you the level of CO2 in the tank, (nb. only a very small proportion of CO2 becomes H2CO3, 99.8% remains as the dissolved gas).
We use bromothymol blue because it is a narrow range pH indicator, yellow at pH6, green at pH7 and blue at pH8. Table below.

Why we need to use 4dKH indicator solution in the drop checker
This is really important, if you had tank water with 20dKH it would naturally be at ~ pH7.8 (at the equilibrium value between HCO3- and atmospheric CO2 levels), but it is almost endlessly carbonate buffered, and if we then added enough CO2 to depress the pH to pH6.8 we would have 94ppm of CO2 and a lot of dead fish.
The Bouncy Castle analogy
If that doesn't make sense and I don't know if this helps, but I'll use the "Bouncy Castle" analogy where "compressed air" substitutes for "added CO2".
When we aren't actively pumping air into the bouncy castle via a compressor, it is deflated and the air pressure inside and outside are the same, this is analogous to a fish tank where we aren't adding CO2.
When we turn the compressor on air is pumped into the castle and it inflates, in the case of CO2, when we add CO2 the HCO3- ~ H2CO3 equilibrium is driven towards H2CO3, you now have an extra H+ ion, and acids are defined as H+ donors, so the pH falls (the water becomes more acid).
If our bouncy castle doesn't have any holes in it, we don't need to add much compressed air to keep it inflated, or in the CO2 case this is the low dKH scenario where a small amount of CO2 addition causes a large pH drop. If the castle has a lot of holes we need to add a lot air to keep it inflated, and in the CO2 case this is the high dKH scenario.
But, rather than estimating how many holes we have, we can have a hole of standard size that always lets out the same amount of air, for CO2 this is the "4dKH solution in the drop checker scenario", where we add a stable amount of air ("CO2") to keep the castle inflated ("CO2 at 25ppm").
In all these cases if we turn the compressed air (CO2) off, the castle deflates and the HCO3- ~ H2CO3 equilibrium returns to its atmospheric level.
Personally for me the air compressor is always going to be turned off, as I can find quite enough ways to accidentally kill my fish without deliberately adding another one.
cheers Darrel
Hi Darrel - what if you're using a PH pen to measure CO2? Does the hardness of the water make a difference here? So in standard (hard) London tap water, what's the CO2 drop that we should be aiming for?
Or have I got the wrong end of the stick entirely? Is it essentially using a drop checker a bit and mainly watching fish to see what the optimum CO2 level is - i.e. the highest we can get away with before fish behaviour is affected - then measuring the PH change of that compared to the water pre-injection, and then using the PH pen over time to adjust rates and try and get it stable / peaking at the right time.
Or have I got the wrong end of the stick entirely? Is it essentially using a drop checker a bit and mainly watching fish to see what the optimum CO2 level is - i.e. the highest we can get away with before fish behaviour is affected - then measuring the PH change of that compared to the water pre-injection, and then using the PH pen over time to adjust rates and try and get it stable / peaking at the right time.
Hi all,
If you use the drop checker 4dKH and bromothymol blue indicator, you don't need to know the pH or hardness of the tank water, you can just use the colour and the pH/kH/CO2 relationship chart to give you a close approximation to the CO2 level. Also because the drop checker is measuring the gas, it isn't effected by other acid forming processes in the tank.
The only downside is that the indicator takes a bit of time to change colour, so it gives you a measure of CO2 level in the past, rather than in real time.
cheers Darrel
Mike it is much easier to use the drop checker. Although both the meter and the and drop checker are measuring pH (the bromothymol blue is a pH indicator), you know the carbonate hardness of the drop checker solution, but not of the tank/tap water.Hi Darrel - what if you're using a PH pen to measure CO2? Does the hardness of the water make a difference here? So in standard (hard) London tap water, what's the CO2 drop that we should be aiming for?
If you use the drop checker 4dKH and bromothymol blue indicator, you don't need to know the pH or hardness of the tank water, you can just use the colour and the pH/kH/CO2 relationship chart to give you a close approximation to the CO2 level. Also because the drop checker is measuring the gas, it isn't effected by other acid forming processes in the tank.
The only downside is that the indicator takes a bit of time to change colour, so it gives you a measure of CO2 level in the past, rather than in real time.
You could use both the pH meter and the drop checker to give you an index for your water, you would measure pH every 10 minutes or so as the CO2 comes on and builds up, and the drop checker changes colour. This would then allow you to just use the pH pen, but it would be more prone to error (if the water had a different hardness to when you performed the calibration run etc.)then measuring the PH change of that compared to the water pre-injection, and then using the PH pen over time to adjust rates and try and get it stable / peaking at the right time.
I'm not a CO2 user, partially because I would always be worried about gassing my fish, but you could observe the fish. I think there are sub-lethal effects to high CO2 exposure, so I would be looking at a lower CO2 level.You could get a fairly quick response from the fish in terms of being more active as the CO2 level drops. The other problem with fish behaviour is that there will be differences between fish species dependent upon the "Root" effect < Fish Respiration>.Is it essentially using a drop checker a bit and mainly watching fish to see what the optimum CO2 level is - i.e. the highest we can get away with before fish behaviour is affected
cheers Darrel
Thanks. Keen to get CO2 right and stable (as I think this helps with Keeping BBA at bay). So I think the right thing to do is:
1. Take PH of water using pen pre-injection
2. Get the CO2 level right with injection using drop checker and fish as guides
3. Measure PH of water throughout photoperiod to see how quickly CO2 level ramps up to ideal rate, how stable it is during photoperiod and how quickly it declines after injection stops
4. Adjust CO2 injection start time, end time (and possibly rate) to make sure it peaks just before lights on and then remains stable until lights off
5. Repeat 1-4 if necessary to fine tune
6. Once sorted, monitor in general using drop checker
1. Take PH of water using pen pre-injection
2. Get the CO2 level right with injection using drop checker and fish as guides
3. Measure PH of water throughout photoperiod to see how quickly CO2 level ramps up to ideal rate, how stable it is during photoperiod and how quickly it declines after injection stops
4. Adjust CO2 injection start time, end time (and possibly rate) to make sure it peaks just before lights on and then remains stable until lights off
5. Repeat 1-4 if necessary to fine tune
6. Once sorted, monitor in general using drop checker
Hi all,
cheers Darrel
That sounds about right.1. Take PH of water using pen pre-injection
2. Get the CO2 level right with injection using drop checker and fish as guides
3. Measure PH of water throughout photoperiod to see how quickly CO2 level ramps up to ideal rate, how stable it is during photoperiod and how quickly it declines after injection stops
4. Adjust CO2 injection start time, end time (and possibly rate) to make sure it peaks just before lights on and then remains stable until lights off
5. Repeat 1-4 if necessary to fine tune
6. Once sorted, monitor in general using drop checker
cheers Darrel
frothhelmet
Member
Hi Darrell, if the air has ~400ppm CO2 currently why does the water only take up ~11ppm in a low tech (this is what it appears to be in the figure above)?
plantbrain
Member
- Joined
- 2 Aug 2007
- Messages
- 1,938
The pH meter works much more accurately than you could possible measure with a drop checker.
Is the Drop Checker(DC) measure lime green, or slightly yellow green?(folks have trouble with this.)
Can I track this color change over the day when the CO2 is being added? (Not really)
How long is the delay between adjusting your CO2 till the DC is measuring what is inside the tank?(couple of hours if things go well)
Is the 4KH reference solution optimal for all tanks? eg, is 30ppm target optimal for all aquariums with CO2? Not even.
While I offered the 1st reference alkalinity solutions for the DIY folks many years ago, I've never been a fan of DC's and I never will be for the above reasons.
pH meters have been effective for the last 20 years for hobbyists if they use them as a relative measure, you can adjust the pH slowly and data log it on your computer.
If you adjust the needle valve just a little bit, you can see a pH change right away if the aquarium has a good flow and good CO2 delivery.
You can adjust say 0.05 pH units down(or up) and observe the fish, plants or algae presence. Watch closely for a week, then add more(or less) or stay the same.
This gives a relative measure of what the CO2 should do to the water's pH, but.........this assumes the KH is stable over that time period, so it must be measured periodically.
If the KH changes, as it can in tap water, then the pH will also change if the CO2 is constant. That's gotten more than one person for sure.
I'm not a fan of pH controllers for that reason.
So the pH meter along with KH measure every so often seems to be the best long term method combined with observation.
I typically use a 1.2 to 1.3 pH drop.
I can then slowly back off till I see any negative growth signs, algae etc.
Same deal with EI dosing, you can slowly back up till you note a negative response.
Then adjust back up to the last prior dosing rate. CO2 is no different.
I really have not found any practical issues with the pH meter method.
Other than the cost........and folks should replace those pH probes once a year or so.
But if you can grow plants well, you can sell some cuttings for trade, cash or credit at the LFS for such items.
Most of my own tanks have 1.2- 1.4 pH drops.
Is the Drop Checker(DC) measure lime green, or slightly yellow green?(folks have trouble with this.)
Can I track this color change over the day when the CO2 is being added? (Not really)
How long is the delay between adjusting your CO2 till the DC is measuring what is inside the tank?(couple of hours if things go well)
Is the 4KH reference solution optimal for all tanks? eg, is 30ppm target optimal for all aquariums with CO2? Not even.
While I offered the 1st reference alkalinity solutions for the DIY folks many years ago, I've never been a fan of DC's and I never will be for the above reasons.
pH meters have been effective for the last 20 years for hobbyists if they use them as a relative measure, you can adjust the pH slowly and data log it on your computer.
If you adjust the needle valve just a little bit, you can see a pH change right away if the aquarium has a good flow and good CO2 delivery.
You can adjust say 0.05 pH units down(or up) and observe the fish, plants or algae presence. Watch closely for a week, then add more(or less) or stay the same.
This gives a relative measure of what the CO2 should do to the water's pH, but.........this assumes the KH is stable over that time period, so it must be measured periodically.
If the KH changes, as it can in tap water, then the pH will also change if the CO2 is constant. That's gotten more than one person for sure.
I'm not a fan of pH controllers for that reason.
So the pH meter along with KH measure every so often seems to be the best long term method combined with observation.
I typically use a 1.2 to 1.3 pH drop.
I can then slowly back off till I see any negative growth signs, algae etc.
Same deal with EI dosing, you can slowly back up till you note a negative response.
Then adjust back up to the last prior dosing rate. CO2 is no different.
I really have not found any practical issues with the pH meter method.
Other than the cost........and folks should replace those pH probes once a year or so.
But if you can grow plants well, you can sell some cuttings for trade, cash or credit at the LFS for such items.
Most of my own tanks have 1.2- 1.4 pH drops.
Hi all,
If you want higher levels of CO2 you need to continually add it. There is a good explanation here: <Aquatic Chemistry>.
cheers Darrel
I've just seen I've never answered this one. The reason is to do with the solubility of gases, but at sea level, at 27oC, pure H2O is saturated with 7.9 mg / l O2, but with only 0.42 mg / l CO2. You can see straight away that CO2 is actually lot more soluble than oxygen, but occurs at a much lower percentage in the atmosphere (400ppm for CO2 & 21% for oxygen).Hi Darrell, if the air has ~400ppm CO2 currently why does the water only take up ~11ppm in a low tech (this is what it appears to be in the figure above)?
If you want higher levels of CO2 you need to continually add it. There is a good explanation here: <Aquatic Chemistry>.
cheers Darrel


