gus6464
New Member
Is this close enough or do I need to get it to 4.0dkh as close as humanly possible? Took me a bit to get to this amount without wanting to make liters of solution.
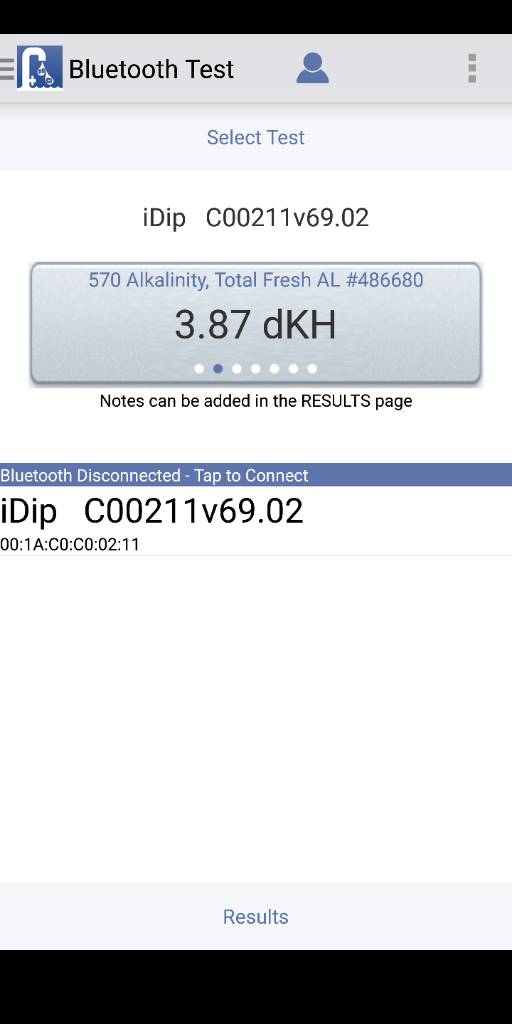

The test kit won't be accurate, but you can make an exact 4dKH from DI water and a soluble carbonate.Is this close enough or do I need to get it to 4.0dkh as close as humanly possible?
Hi all,The test kit won't be accurate, but you can make an exact 4dKH from DI water and a soluble carbonate.
Sodium bicarbonate (NaHCO3) is usually used, but sodium carbonate (Na2CO3), potassium bicarbonate (KHCO3) and potassium carbonate (K2CO3) are other options.
The workings for NaHCO3 are in this post <"4dKH Solution">.
cheers Darrel
I'm not sure what this statement meansThat sensafe is a commercial tester.
That sensafe is a commercial tester. I have compared the alkalinity results with a saltwater sample sent to an ICP-OES lab when I had my reef tank and it was within 0.05 dkh. I made the solution with RODI water and baking soda.
@alto is a scientist, and we are naturally sceptical.I looked at the SenSafe website but there are no (recognized) certifications (of the photometer) or specifications of product performance in terms of accuracy or precision, no mention of any calibration check etc
I'm not sure about that one, <"ICP"> is used to measure metal ions from their emission spectra in very hot plasma. It would tell you the exact amount of Na+, Mg++, Ca++ ions in your sea water sample, but you would then have to use a formula to estimate the dKH.I have compared the alkalinity results with a saltwater sample sent to an ICP-OES lab when I had my reef tank and it was within 0.05 dkh.
Hey, i made the 4dkh solution, is it bad if i add more than 1 drop of ph liquid test ? thxHi all,@alto is a scientist, and we are naturally sceptical.
It probably will be pretty close, because the test meter uses photometry, although the fixed wave length (570nm ~ yellow/green light) may mean that you have a non-standard analytical method, and the cell is still measuring the colour of a test strip (blue?).
Because the iDip test is for "total alkalinity" the test strip has "performed" an <"acid base titration">, and all the hardness in your sample is carbonate "temporary" hardness (you only have NaHCO3 as a base), which we express as dKH.
I would go exactly the other way around, because you have the "baking soda" (pure NaHCO3) and DI water, you can test your IDip.
You need to start with dry NaHCO3, you can dry it in a low temperature oven (350oF (~175oC) will do), and you will probably find you get about 7g of dry NaHCO3 if you start with 10g (should take about half an hour to fully dry out).
After that 30mg (0.03g) NaHCO3 in 1 litre gives 1dKH and 120mg (0.12g) in 1 litre gives 4dKH. This uses the workings from <"the Krib: water hardness">
Because I don't like working with small weights, or volumes, of chemicals, I'd use 6g of NaHCO3 in 500cm3, which is a 400dKH stock solution.
You can store the 400dKH solution in a stoppered bottle for as long as you like.
If you take 5cm3 of this stock (which I'd weigh as 5g) and make it up to 500cm3 (500g) with DI, you have a 4dKH solution, to which you add a drop of <"bromothymol blue>". I'm not sure about that one, <"ICP"> is used to measure metal ions from their emission spectra in very hot plasma. It would tell you the exact amount of Na+, Mg++, Ca++ ions in your sea water sample, but you would then have to use a formula to estimate the dKH.
If any-one is interested we are still trying to purchase an ICP, at the moment we are arguing about who pays for the argon (Ar). We have an AAS, but that uses acetylene and argon is a lot safer (which is why we may get the money for the ICP).
cheers Darrel
It shouldn't matter, it is a weak acid but you need to have enough <"bromothymol blue"> to be able to see the pH colour change.Hey, i made the 4dkh solution, is it bad if i add more than 1 drop of ph liquid test ?
