-
You are viewing the forum as a Guest, please login (you can use your Facebook, Twitter, Google or Microsoft account to login) or register using this link: Log in or Sign Up
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
tapwater contains nitrates?
- Thread starter Mitchel
- Start date
MichaelJ
Member
Hi @Mitchel Did you get the nitrate (NO3) reading from a water report or did you measure it yourself (measuring NO3 can be quite difficult with a test kit) ? Even if you know for sure its 15 ppm out of your tap, I would be cautious relying on "fertilizers" from tap water as it may vary quite a bit throughout the year. Personally, I would disregard it and just create a stock solution not taking the tap water NO3 level into account. Alternatively, you could factor it in a bit by just going a smidge lower with the NO3 with the stock solution than you otherwise would.If I want to make my nutrients I use dry salts.
But my tapwater also contains nitrates. (15ppm) Should I take this into account when creating a stock solution?
This is because I do a 50% weekly water change with tap water
Tia, Mitch
Cheers,
Michael
I think it's a bit tricky because I want to have a 'lean-dose' approach.
My target would be approx 7 ppm nitrates so I'll would have to work with 50% osmosis/tap water.
So I wouldn't have to use ferts for the NO3 because it's already present in my tap water (according to my water company)
My target would be approx 7 ppm nitrates so I'll would have to work with 50% osmosis/tap water.
So I wouldn't have to use ferts for the NO3 because it's already present in my tap water (according to my water company)
Can you post your water report here?If I want to make my nutrients I use dry salts.
But my tapwater also contains nitrates. (15ppm) Should I take this into account when creating a stock solution?
This is because I do a 50% weekly water change with tap water
Tia, Mitch
Can you not go RO all the way? This will in turn allow you to do whatever you want in terms of ferts without having to wonder how to reach a target that you will never reach simply because salt concentration in your tap water can and will inevitably vary during the course of the year.I think it's a bit tricky because I want to have a 'lean-dose' approach.
My target would be approx 7 ppm nitrates so I'll would have to work with 50% osmosis/tap water.
So I wouldn't have to use ferts for the NO3 because it's already present in my tap water (according to my water company)
Hi all,
Any of the plant nutrients can be <"Liebig's limiting nutrient">, but as long as all <"fourteen essential elements"> are present, <"leaf colour and growth"> are closely linked to nitrogen availability.
cheers Darrel
I'd just use a <"complete fertiliser"> (including nitrogen (N)), but don't have a "target value" as such. You can use the plants to deplete the nitrate (NO3-) in the tap water and then use the <"Duckweed Index"> as an indication of when to add more fertiliser.I think it's a bit tricky because I want to have a 'lean-dose' approach.
My target would be approx 7 ppm nitrates
Any of the plant nutrients can be <"Liebig's limiting nutrient">, but as long as all <"fourteen essential elements"> are present, <"leaf colour and growth"> are closely linked to nitrogen availability.
cheers Darrel
Can you post your water report here?
Attachments
Hi all,
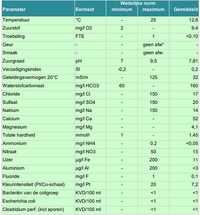
That looks fine. The 160 mg / L bicarbonate (HCO3-) is about 7dKH (<"1dKH = 21.8 ppm (mg / L) HCO3-">) and the dGH about the same, from the calcium value of 52 mg / L (1dGH = 7.14 ppm Ca++) and 4 ppm Mg. Have a look at <"Some handy facts about water">.
I'd be pretty happy with that as a starting point, the pH just reflects the 2HCO3- content and the conductivity is about 320 microS / cm.
cheers Darrel

That looks fine. The 160 mg / L bicarbonate (HCO3-) is about 7dKH (<"1dKH = 21.8 ppm (mg / L) HCO3-">) and the dGH about the same, from the calcium value of 52 mg / L (1dGH = 7.14 ppm Ca++) and 4 ppm Mg. Have a look at <"Some handy facts about water">.
I'd be pretty happy with that as a starting point, the pH just reflects the 2HCO3- content and the conductivity is about 320 microS / cm.
cheers Darrel
But actually I find it strange.
If you make your own nutrients, you can never achieve exact targets when you work with tap water. Hypothetically speaking, would a 'lean-dose' approach only be efficient if you work with RO water?
Can I then conclude that it does not actually have to be calculated so precisely,and that an approximation of a given ppm must be seen in a bigger perspective.
e.g. 15 ppm is perfect, but 10 or 20 is also good?
But when I look at my commercial bottle of nutrition and it says: with every 10ml addition you get 15ppm nitrate in the water.
Fine, but if my tap water also contains 15 ppm, so I suddenly have 30 ppm in the water.
Same situation with the EI. Average targets for nitrate are 22.5 ppm. So if I perform a water change, 15ppm comes out of the tap and according to the EI, I get a total of 37.5ppm nitrate in my tank.
I know it's not all that important, and that you 'reset' everything with a weekly water change. But I find it very confusing when people say: your tank needs (as an example) 22,5ppm nitrates.
But in reality you have much higher values as your tabwater has 'nutrients' in it...
If you make your own nutrients, you can never achieve exact targets when you work with tap water. Hypothetically speaking, would a 'lean-dose' approach only be efficient if you work with RO water?
Can I then conclude that it does not actually have to be calculated so precisely,and that an approximation of a given ppm must be seen in a bigger perspective.
e.g. 15 ppm is perfect, but 10 or 20 is also good?
But when I look at my commercial bottle of nutrition and it says: with every 10ml addition you get 15ppm nitrate in the water.
Fine, but if my tap water also contains 15 ppm, so I suddenly have 30 ppm in the water.
Same situation with the EI. Average targets for nitrate are 22.5 ppm. So if I perform a water change, 15ppm comes out of the tap and according to the EI, I get a total of 37.5ppm nitrate in my tank.
I know it's not all that important, and that you 'reset' everything with a weekly water change. But I find it very confusing when people say: your tank needs (as an example) 22,5ppm nitrates.
But in reality you have much higher values as your tabwater has 'nutrients' in it...
Hi all,

You know you have plenty of fixed nitrogen, rapidly being converted into plenty of plant tissue.
cheers Darrel
No, not really, you can use lean dosing with any starting water, you just need to <"watch the plants, they can't lie">.If you make your own nutrients, you can never achieve exact targets when you work with tap water. Hypothetically speaking, would a 'lean-dose' approach only be efficient if you work with RO water?
Yes, exactly that. It isn't that I'm not interested in water parameters, <"I'm very interested">, but I also realise that <"there are mechanisms"> you can use to give you a ball-park figure, and that <"ball-park"> is near enough.Can I then conclude that it does not actually have to be calculated so precisely,and that an approximation of a given ppm must be seen in a bigger perspective.
e.g. 15 ppm is perfect, but 10 or 20 is also good?
But only briefly, until the plants deplete it. So realistically that number is always shrinking until you add some more NO3-, either from your water change or from your fertiliser.But when I look at my commercial bottle of nutrition and it says: with every 10ml addition you get 15ppm nitrate in the water.
Fine, but if my tap water also contains 15 ppm, so I suddenly have 30 ppm in the water.
But ~no-one actually knows <"how much nitrate they have"> and the <"needs of plants will differ">, dependent <"upon their growth rate etc">. If you <"have these, (belonging to @Cédric )Same situation with the EI. Average targets for nitrate are 22.5 ppm. So if I perform a water change, 15ppm comes out of the tap and according to the EI, I get a total of 37.5ppm nitrate in my tank.
I know it's not all that important, and that you 'reset' everything with a weekly water change. But I find it very confusing when people say: your tank needs (as an example) 22,5ppm nitrates.
You know you have plenty of fixed nitrogen, rapidly being converted into plenty of plant tissue.
cheers Darrel
MichaelJ
Member
Hypothetically speaking, would a 'lean-dose' approach only be efficient if you work with RO water?
There are plenty of wiggle room. You can grow plants in almost any type of water suitable for human consumption - you may face some limitations on plant choices however.. Looking at your water report - which looks pretty good - I wouldn't really worry about it too much as long as you just keep in mind that the parameters likely will change over time due to seasonal variations, how the water company source and treat the water etc. Everyone using tap water - which are most I believe - are faced with this and its usually not an issue. You watch the plants and adapt. Another thought is that lots of all this depends on what you’re trying to accomplish, your individual skill level , what plants you’re planning to grow and what level of maintenance complexity you are prepared to deal with... As @Hanuman said, and since your already vested in RO (I assume), why not just go 100% RO - which should be manageable with your 80L tank (which seems to be the size I picked up from a prior post) and you can disregard all concerns about trying to factor in whatever is in your tap water... Just a thought.
Cheers,
Michael
Last edited:
KirstyF
Member
I think the answer, unfortunately, is it depends.
If there were one single approach to ferts, we’d have alot less to talk about here. 😂
EI is an everything in excess approach so 20pm, 30ppm …….does it really matter….not really, so long as you have more than the plants need. Super easy, especially for folks newer to the hobby. Can result in pretty rapid growth and a good bit of trimming needed.
A ‘lean’ approach probably invites more precision and people may be more specific regarding PPM’s and even ratio’s of the various nutrients in their tanks. Can be more complex, less margin for error, but can also offer great results.
A Duckweed approach is, watch the plants and add more ferts if they look like they need it. Again, super simple. Requires good observation and an understanding of what you are seeing.
I’m probably (definitely) oversimplifying some of this but you get my drift. 🤔
Also, just to make it more fun, many plants will respond differently in harder water by comparison to softer water, and different plants have different needs. I can grow Vallisneria in my hard water, like it’s going out of fashion, but have yet to get Wallichii to take at all.
So……I’ve made the decision to work with my tap water (700 ltd, RO is not an option) which has at least 20-30ppm of Nitrates in it and I therefore no longer dose any additional. With full RO, you can specify whatever you want. With half and half, you can ignore the tap and dose EI style or you can work with the tap and reduce accordingly to a leaner target, but you will always get a little variation.
Ultimately your plants will tell you what is working and what isn’t but reading them is a whole skill set in itself.
I personally quite like to tinker so never make up more than 3 weeks ferts at a time. That way I can adjust as I feel I need to, without throwing good ferts away.
I think the first thing you have to figure out is what approach you prefer and why?
If there were one single approach to ferts, we’d have alot less to talk about here. 😂
EI is an everything in excess approach so 20pm, 30ppm …….does it really matter….not really, so long as you have more than the plants need. Super easy, especially for folks newer to the hobby. Can result in pretty rapid growth and a good bit of trimming needed.
A ‘lean’ approach probably invites more precision and people may be more specific regarding PPM’s and even ratio’s of the various nutrients in their tanks. Can be more complex, less margin for error, but can also offer great results.
A Duckweed approach is, watch the plants and add more ferts if they look like they need it. Again, super simple. Requires good observation and an understanding of what you are seeing.
I’m probably (definitely) oversimplifying some of this but you get my drift. 🤔
Also, just to make it more fun, many plants will respond differently in harder water by comparison to softer water, and different plants have different needs. I can grow Vallisneria in my hard water, like it’s going out of fashion, but have yet to get Wallichii to take at all.
So……I’ve made the decision to work with my tap water (700 ltd, RO is not an option) which has at least 20-30ppm of Nitrates in it and I therefore no longer dose any additional. With full RO, you can specify whatever you want. With half and half, you can ignore the tap and dose EI style or you can work with the tap and reduce accordingly to a leaner target, but you will always get a little variation.
Ultimately your plants will tell you what is working and what isn’t but reading them is a whole skill set in itself.
I personally quite like to tinker so never make up more than 3 weeks ferts at a time. That way I can adjust as I feel I need to, without throwing good ferts away.
I think the first thing you have to figure out is what approach you prefer and why?



