Day 08 update: Water change 50 %.
-
You are viewing the forum as a Guest, please login (you can use your Facebook, Twitter, Google or Microsoft account to login) or register using this link: Log in or Sign Up
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Maq's experiment 23b
- Thread starter _Maq_
- Start date
-
- Tags
- algae fertilization
Wookii
Member
Day 08 update: Water change 50 %.
Do we have an updated image of the tanks?
EDIT: Sorry I missed this:
Probably on Tuesday I'll make new set of pics, and post them here, of course.








Most growth defects are poorly visible on pics, so here's the description:
Rotala wallichii - not growing (stunted?) in C, D.
Bacopa lanigera - chlorosis on top leaves in A, B, less pronounced in C.
Hygrophila corymbosa - chlorosis on top leaves in A.
Ammannia pedicellata - stunted in D, deformed in C.
Last edited:
@_Maq_ Can you please put a Text on each picture indicating which is which?
I can see the name of the pic when I click on it. The names are made to be pretty descriptive, do you need to explain D(10).jpg?put a Text on each picture
Ah I see, I didn't notice the names on each pictures earlier.I can see the name of the pic when I click on it. The names are made to be pretty descriptive, do you need to explain D(10).jpg?
MichaelJ
Member
Most growth defects are poorly visible on pics, so here's the description:
Rotala wallichii - not growing (stunted?) in C, D.
Bacopa lanigera - chlorosis on top leaves in A, B, less pronounced in C.
Hygrophila corymbosa - chlorosis on top leaves in A.
Ammannia pedicellata - stunted in D, deformed in C.
Of course, way, way too early and premature to comment, so here goes....
Cheers,
Michael
Last edited:
John q
Member
I'm not even thinking of commenting on the progression of the plants in either tank at this stage. The obvious thing that does stand out is the amount of algae on the substrate in tank B and to a slightly lesser extent tank A. Will be interesting to see how that develops.Of course, way too early to comment
Edit: to be fair tank B started out with slightly more algae looking at original pics.
MichaelJ
Member
True. It bugs me that the tanks weren't cleaned up before the experiment started - especially since one of the objectives is to figure out the correlation between NH4 and algae....The obvious thing that does stand out is the amount of algae on the substrate in tank B and to a slightly lesser extent tank A.
Cheers,
Michael
Yes. Each of the tanks has got a different history prior to this test. So, algae, par example, must be compared to initial state.to be fair tank B started out with slightly more algae looking at original pics.
I understand. My previous practice with these experiments and these tanks tells me this: The algae are basically the same in all of them. Sometimes there's more of them in A, other times elsewhere. We're not going to make conclusions after one or two weeks. If any of the tanks contains "something" that causes algae outbreak, we'll see it clearly, sooner or later. So far, algae are within ranks of casual oscillation. Believe me, I run these tanks quite a while.It bugs me that the tanks weren't cleaned up before the experiment started
Wookii
Member
True. It bugs me that the tanks weren't cleaned up before the experiment started - especially since one of the objectives is to figure out the correlation between NH4 and algae....
Cheers,
Michael
Me too - that should really have been a primary requirement @_Maq_ especially given the simplicity of achieving it.
The algae levels in most of the tanks would be considered unacceptable to many on this forum, and I would consider tanks A and B as having an algal bloom had I seen them in isolation, but now it is much more difficult to identify if we are just seeing further growth of existing algae.
Andy Pierce
Member
Yes, and what you'd do if the algae was in your tank would be to scrape it off. That can still happen at some common timepoint coming up and then the algae regrowth (or lack of regrowth) can be reasonably monitored. For me the regrowth is much more relevant than initial growth from a totally clean state.The algae levels in most of the tanks would be considered unacceptable to many on this forum, and I would consider tanks A and B as having an algal bloom had I seen them in isolation, but now it is much more difficult to identify if we are just seeing further growth of existing algae.
Yes, that works for one individual tank, but provides little improvement in terms of comparing the tanks/ treatments between each other, as Wookii mentions. Three of the four tanks have the same filter... we can compare. But each of the four started at different algae levels, a variable that impacts directly the algae growth but also indirectly the plant growth... comparison is less objective.
A simple scraping off the glass won’t equalize the impact done the previous weeks and arguably neither will it equal the amount of algae present in the tanks. In one tank, scrape all 10000 algae, do a 90% water change you are left with 1000 algae in the water for regrowth. In another tank, scrape all 100 algae, do a 90% water change you are left with 10 algae in the water for regrowth. Again the regrowth starts on unequal footing and it gets worse with only 50% water change as planned.
Given Maq‘s disposition in his recent posts, I think it‘s only fair to take the journal updates for these four tanks as they come. Four tank journals in one thread is still an interesting read.
A simple scraping off the glass won’t equalize the impact done the previous weeks and arguably neither will it equal the amount of algae present in the tanks. In one tank, scrape all 10000 algae, do a 90% water change you are left with 1000 algae in the water for regrowth. In another tank, scrape all 100 algae, do a 90% water change you are left with 10 algae in the water for regrowth. Again the regrowth starts on unequal footing and it gets worse with only 50% water change as planned.
Given Maq‘s disposition in his recent posts, I think it‘s only fair to take the journal updates for these four tanks as they come. Four tank journals in one thread is still an interesting read.
I'm afraid the pics provide somehow distorted view. I can see it in real and would not call the state of any of the tanks "algal bloom". Perhaps you should look at the back wall. Side walls reflect the green of the plants.I would consider tanks A and B as having an algal bloom
Neither tank is crystal clear. Never was. You can see algae on the substrate, too. In some tanks. Those I cannot remove without disturbing substrate => microbial community.
The only way to make tanks equal would be to start from scratch and cycle them. That would be impractical, and distorting the results as well, just in another way.
Please, don't be too strict with me. I think a perfect test would require management and tools on higher level. I must make some compromises. So far, the test runs for 12 days only. We'll see more relevant results during weeks to come.
EDIT: Filters. There's no observable difference in the flow of water all of them produce.
Last edited:
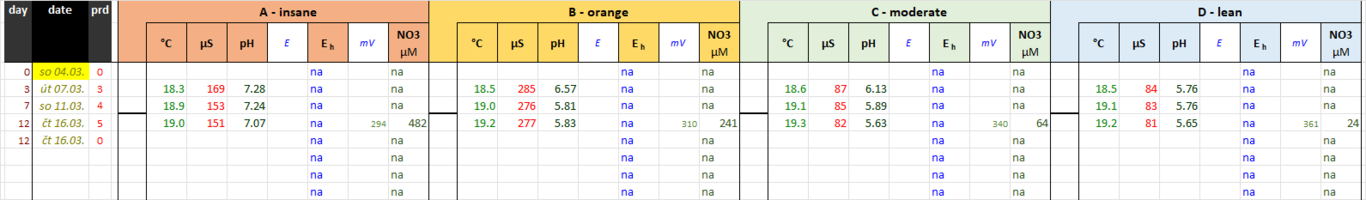
Some notes related to NO3 measurement:
(1) My ISE electrode is several years old and I can't calibrate it. It's precision is therefore compromised. Still, it regularly renders data within the range of "fairly possible", so it's probably not wildly off-mark.
(2) Ammonium adsorbs well on sand and especially on detritus. If you change 50 % of water, you don't remove 50 % of ammonium in the tank; probably much less. Ammonium may accumulate and you can't measure it. On the other hand, nitrates do not adsorb on anything. In this way you can sometimes detect nitrates in concentration higher than any concentration of ammonium you dosed before.
(3) Note that nitrification is running fairly effectively without any filtration, not to mention advanced media featuring huge colonization area.
Today, Day 13, dosing micros:
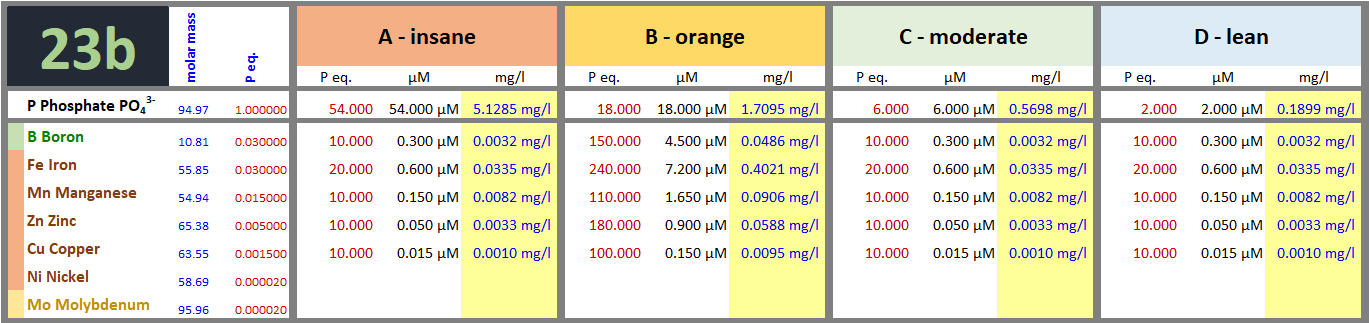
Compared to previous, I've doubled the dose of iron in tanks A, C, D. I'd like very much to avoid iron deficiency - not to meddle in detecting possible magnesium deficiency.
Algae are strengthening in tanks A, B, C to a degree that they begin to get visibly attached on plants. That's my personal limit between harmless and unacceptable algae growth.
Compared to previous, I've doubled the dose of iron in tanks A, C, D. I'd like very much to avoid iron deficiency - not to meddle in detecting possible magnesium deficiency.
Algae are strengthening in tanks A, B, C to a degree that they begin to get visibly attached on plants. That's my personal limit between harmless and unacceptable algae growth.
Wookii
Member
View attachment 202513
Some notes related to NO3 measurement:
(1) My ISE electrode is several years old and I can't calibrate it. It's precision is therefore compromised. Still, it regularly renders data within the range of "fairly possible", so it's probably not wildly off-mark.
(2) Ammonium adsorbs well on sand and especially on detritus. If you change 50 % of water, you don't remove 50 % of ammonium in the tank; probably much less. Ammonium may accumulate and you can't measure it. On the other hand, nitrates do not adsorb on anything. In this way you can sometimes detect nitrates in concentration higher than any concentration of ammonium you dosed before.
(3) Note that nitrification is running fairly effectively without any filtration, not to mention advanced media featuring huge colonization area.
That's interesting - so that's quite a lot of NO3, even in the 'leanest' tank (D)? And you've dosed no NO3 to any tank is that right, so all the NO3 exists through nitrification of the dosed ammonia by the substrate? - if so, that's a very interesting observation irrespective of the other experiment objectives. It's not something I've considered before, but a quick google suggests that 1ppm ammonia results in 3.6ppm NO3 after nitrification - is that right?
Are you able to take measurements of the water column ammonia levels with your kit?
Last edited:
No nitrate dosing. 1 ppm ammonia is 3.44 ppm NO3. But beware the units - it's µM. ... 16 µM NO3 = 1 mg/l NO3.you've dosed no NO3
I see nothing surprising in nitrification running that quickly. See my signature: no filters. Not even @dw1305 with his disgust for marketing dishonesty has as yet gone as far as to admit the redundancy of filters (in most cases); he's only dismissed highly porous media so far, if I'm not mistaken.
Don't even think of me measuring ammonia!
Hi all,
<"I still have a filter">, although when I used to have internal filters I used to regularly <"fail to turn them on after cleaning"> without any issue.
If it was a binary choice, where you could only have one or the other? I would always rather have plants (so really always "plants & microbes") than a filter, but I understand there are situations where <"the filter (and filter media) become more important">.
cheers Darrel
I think we both have the same view about what is really important , and that is maintaining a high <"level of dissolved oxygen">. For me<"it is the coffee"> and everything else is <"really just froth">.Not even @dw1305 with his disgust for marketing dishonesty has as yet gone as far as to admit the redundancy of filters (in most cases); he's only dismissed highly porous media so far, if I'm not mistaken.
<"I still have a filter">, although when I used to have internal filters I used to regularly <"fail to turn them on after cleaning"> without any issue.
If it was a binary choice, where you could only have one or the other? I would always rather have plants (so really always "plants & microbes") than a filter, but I understand there are situations where <"the filter (and filter media) become more important">.
cheers Darrel


