Zeus.
Fertz Meister
Hi all,
Myself and @Hanuman are in the process of adding a more dedicated Remineralising sheet to the IFC Calculator so folk can make their own remineralisers at a fraction of the cost, with dry dosing and/or remineralising solutions which suit smaller tank remineralising also IMO. It also makes it clear which salts are not suitable for remineralising solutions also due to their limited solubility.
(alpha preview below)
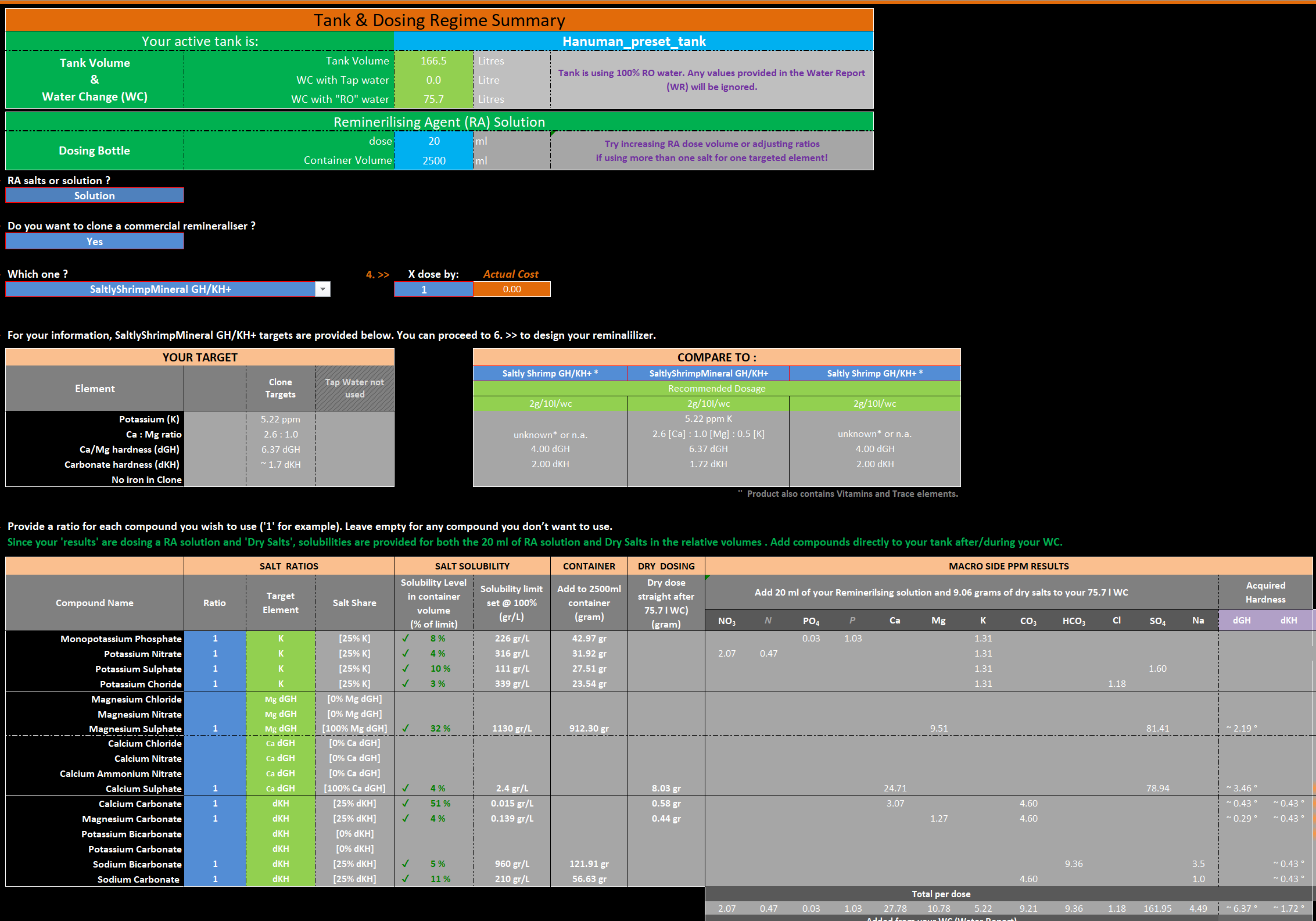
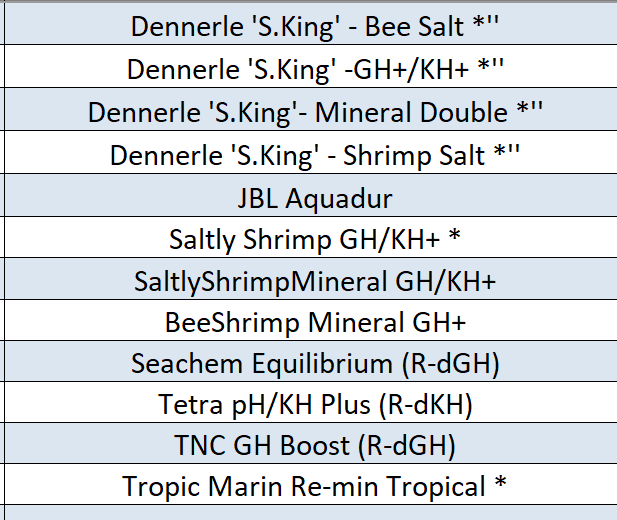
Much has been done and still lots to do. We do have a reasonable list of commercial remineralisers below, if your using one that's not on the list please post and let us know so we can add them to our database for comparing/cloning
Cheers Zeus.
Data sheet/PDF or pic of data on back of product please, link to website 'if' it there is one -please
Myself and @Hanuman are in the process of adding a more dedicated Remineralising sheet to the IFC Calculator so folk can make their own remineralisers at a fraction of the cost, with dry dosing and/or remineralising solutions which suit smaller tank remineralising also IMO. It also makes it clear which salts are not suitable for remineralising solutions also due to their limited solubility.
(alpha preview below)
Much has been done and still lots to do. We do have a reasonable list of commercial remineralisers below, if your using one that's not on the list please post and let us know so we can add them to our database for comparing/cloning
Cheers Zeus.
Data sheet/PDF or pic of data on back of product please, link to website 'if' it there is one -please
Last edited:




 which would convert 2 e KH to 2.3 dKH
which would convert 2 e KH to 2.3 dKH