-
You are viewing the forum as a Guest, please login (you can use your Facebook, Twitter, Google or Microsoft account to login) or register using this link: Log in or Sign Up
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Got quite a few of mine direct from Solufeed eg Fe EDDHA price soon adds up all the same, others I used small amounts from e-bay
Molybdenum Ammonium Heptamolybdate Pure (NH4)6Mo7O24 Planted Aquarium Fertilizer
Molybdenum Ammonium Heptamolybdate Pure (NH4)6Mo7O24 Planted Aquarium Fertilizer
others I used small amounts from e-bay
Yes that would be my last resort if I can't find locally at a reasonable price. Buying from ebay always carries the risk of the product not being the grade you want and there is no way one can check the quality without a lab to test the product.
If I only had plants and no fish I wouldn't worry too much and probably would buy the technical grade. Buying lower grades may carry the risk of those salts containing undesirable levels of heavy metals, or so I was told.
Last edited:
Well well I just talked to 5 different producers of liquid fertilizers (2 local and 3 international that are well established) for planted aquarium and they all told me the same in regard to salts grade. They use glasshouse/agricultural/factory grades. No need for lab grade salts as the difference in purity is marginal to the point where it makes no difference in the tank.
Below is the analysis of every traces (technical grade) I asked a quote for. If any knowledgeable person in the area of chemistry has something to say before I go out and buy these traces now it's the time 😉








Thanks for all the help guys.
Below is the analysis of every traces (technical grade) I asked a quote for. If any knowledgeable person in the area of chemistry has something to say before I go out and buy these traces now it's the time 😉








Thanks for all the help guys.
Last edited:
Well expecting (hoping) a positive answer I went out and got the traces. Now I got enough to make liquid fertilizer for 3 generations to come perhaps even 4 or 5  . Still have the potassium sorbate and the ascorbic acide to buy but those I will buy from a different supplier (100 grams) as the supplier where I bought the other traces only sold 1kg minimum and those chemical were somehow expensive for whatever reason.
. Still have the potassium sorbate and the ascorbic acide to buy but those I will buy from a different supplier (100 grams) as the supplier where I bought the other traces only sold 1kg minimum and those chemical were somehow expensive for whatever reason.


Last edited:
You don't have to use a Gluconated Iron source, I like to use it because of its low persistence and that it comes mixed with Humic Acid which may provide some natural chelation for the unchelated traces when it all gets mixed up in the bottle. My aim is to have zero EDTA in the tank as it won't play nice with the Fe and let go of it all at the higher KH/pH I run my tank. I boost my Fe Gluconate dose with FeDTPA (11%) instead as it has a little better persistence at higher pH (detectable the day after dosing).
If I don't add a Gluconated Iron source what are my options? Presently I have these possible iron sources available:
Iron DTPA (Fe-DTPA 7%)
Iron EDTA (Fe-EDTA 13.5%)
As I was going to follow somewhat Jame's recipe and yours, I also bought C6H8O6 (Ascorbic Acid). Would this be ok to replace the Gluconated Iron and instead increase the dose of either Fe-DTPA 7% or Fe-EDTA 13.5%?
An alternative to the Microbe-Lift Plants Fe for ease of use if you can obtain it would be Seachem Flourish Iron (not Flourish Comprehensive as that contains low doses of trace elements).
Yes it can be found here but at 15USD the 100ml bottle. Since you are adding 50ml, that's basically 7.5USD just in Seachem Iron for the 500ml total solution. At that price I can already buy myself a 500ml of pre-made Iron trace from one of the liquid fertiliser producers here. The whole point of this DIY stuff is to make it cheaper.
If you have trouble sourcing the Seachem Iron then an alternative is to source Ferrous Gluconate itself, it can can come as a powder or as a liquid and you may be able to get this from a Pharmacy, I would go for the powder if you can. You would also need to source Humic Acid (as a dry powder if you can) and mixing in water (RO/DI or distilled water for zero impurities) with the Fe Gluconate should give you a Seachem or Microbe-Lift facsimile.
I found this: https://www.lazada.co.th/products/h...o4m.searchlist.list.7.15093045nOXVlQ&search=1
Not much info on the spec of that Humic Acid other than it's 80%. For Ferrous Gluconate strangely I haven't been able to find online locally. I will see if in a pharmacy they sell.
The choice between using DTPA or EDTA for the Iron depends on the pH of the tank, if you are below 6.5 most of the time then EDTA would do but if the pH is any higher then it will start dropping the Iron making it unavailable. I have measured lower than half of my DTPA dose the next day having spent daytime at pH 6.5 and overnight around pH 7.8 (my KH is about 7.8-8).
I measured my water PH prior Co2 injection and PH was at ~ 7.0 / 7.1. During Co2 injecting PH drops to ~ 6.4 / 6.5. I suppose I would be better off with Fe-DTPA 7%, correct?
Last edited:
X3NiTH
Member
- Joined
- 13 Apr 2014
- Messages
- 1,669
Yes at that pH DTPA is the better Iron chelate.
I should add that you want Fulvic acid in addition to the Humic acid. I have to admit that I have not reached this part of the process for blending this mixture, I have not yet sourced the base salts myself, the ones I had looked at were made by NPK-Industries, the Humic Acid they supply is sourced from Leonardite (there are sources for this in Thailand, here is an analysis of such deposits), the product I specifically looked at was their Full-Up which was Humates with 10% Humic Acid, the humates in this case will contain a decent proportion of Fulvic Acid, have a look at the MSDS for more detail. The K component in the Humic Acid is because the Humic Acid is only recoverable in Alkali water and they have used Potassium Hydroxide as the moderator. Fulvic Acid is fully soluble in water.
You can DIY the Humates using peat, I'm sure there will be a detailed guide out there to do this but I found some instructions in the preview of a related Technical Journal on JSTOR.
I should add that you want Fulvic acid in addition to the Humic acid. I have to admit that I have not reached this part of the process for blending this mixture, I have not yet sourced the base salts myself, the ones I had looked at were made by NPK-Industries, the Humic Acid they supply is sourced from Leonardite (there are sources for this in Thailand, here is an analysis of such deposits), the product I specifically looked at was their Full-Up which was Humates with 10% Humic Acid, the humates in this case will contain a decent proportion of Fulvic Acid, have a look at the MSDS for more detail. The K component in the Humic Acid is because the Humic Acid is only recoverable in Alkali water and they have used Potassium Hydroxide as the moderator. Fulvic Acid is fully soluble in water.
You can DIY the Humates using peat, I'm sure there will be a detailed guide out there to do this but I found some instructions in the preview of a related Technical Journal on JSTOR.
Man this is becoming more of an alchemist experiment than making a fertiliser. Don't take it wrong, I am laughing about it! Obviously this isn't as simple as many people claim it is and I am starting to understand why many end up just buying the pre made stuff. Lots of headache. In fact this DIY fertilizer stuff is really not for the beginner or those with say only one tank or two. Not worth the pain unless you can source the CSM+b and the macros easily. Perhaps most people can but I find it more interesting to do something that doesn't involve depending on some mixtures with artificial chelating agent.
Since I bought most of the chemicals now perhaps I should pursue the holly grail of a fully non-synthetic chelating agent recipe (although the fact is I already posses all the chelated traces as well, but as you said it would involve a lot of math so I abandoned the idea of using those). Perhaps we can come up with a recipe that does not require any pre-mixed products (like Microbe-Lift Plants Fe, Seachem Flourish Iron, CSM + B or other stuff out there). It would be great to be able to make a fertilizer that is fully composed of non pre-chelated individual ingredients. Perhaps that's not even possible. I guess the most difficult part would be to make a solution that is stable in time.
Since I bought most of the chemicals now perhaps I should pursue the holly grail of a fully non-synthetic chelating agent recipe (although the fact is I already posses all the chelated traces as well, but as you said it would involve a lot of math so I abandoned the idea of using those). Perhaps we can come up with a recipe that does not require any pre-mixed products (like Microbe-Lift Plants Fe, Seachem Flourish Iron, CSM + B or other stuff out there). It would be great to be able to make a fertilizer that is fully composed of non pre-chelated individual ingredients. Perhaps that's not even possible. I guess the most difficult part would be to make a solution that is stable in time.
Last edited:
Looking around locally I was able to find some suppliers of humic and fulvic acid. The humic acid product contains usually 80% of it and the fulvic acid, 62%. Problem is they don't even provide an analysis of the products so impossible to check what are the remainders.
X3NiTH
Member
- Joined
- 13 Apr 2014
- Messages
- 1,669
Yeah it does feel a little like Alchemy, a little bit of this and a little bit of that, except it's in the pursuit of turning the elements into healthy plant tissue. I like to think I am helping to counteract the Magic that Bucephalandra use to do a vanishing act when they don't like what's on offer, this from a plant that supposedly thrives on thin gruel (Rainwater over Karst and roots in Interstitial Soils which are mainly composed of dead epiphytes). From what I have seen they appear to prefer their breakfast wrapped in a tasty pastry they can absorb for extra nutrition rather than have them offered it wrapped in a synthetic they can't absorb, and I fear if they do manage to tear up that synthetic wrapper and if it is Ethylene based could be a cause to trigger tissue loss wherever it was dismantled (ethylene in a plant is used as a signalling hormone to trigger cell death). Also I remember reading something somewhere (can't remember where) that one of the problems of using the wrong synthetic chelate for the pH range of the target element is that it could lead to unbound chelate being absorbed into the plant where it then chelates with something the plant wants to store and then robs it of nutrients if it gets transported back out the plant. As a consequence of the pH range I run at I'm keeping away from EDTA traces and only using a small amount of FeDTPA for Iron persistence day to day.
It's quite interesting to compare the size difference between the Humic and Fulvic Acid molecules. Shortcrust pastry or Puff pastry, I assume they both appear quite delicious from a plants perspective.

This is quite an interesting article - Humates and Humic Acid. How do they work?
Commenting on the purity of the Humic and Fulvic Acid sources you found it might be worth comparing.
Here's the back label for the RAW brand Humic Acid source I mentioned above.

As you can see it's sourced from Leonardite and it is only 59% purity, so your 80% purity grade appears to be a more concentrated product, very likely it is from a locally sourced Leonardite deposit, the question you need to ask is how it was prepared, which hydroxide was used to solvate the Humic Acid, the RAW one above uses Potassium Hydroxide hence the 4% K content, other hydroxides that can be used are Sodium Hydroxide or Ammonium Hydroxide (from what I've read the Ammonium formulation smells pretty bad). I would prefer the Potassium based product simply because it's the preferred transport ion within plants, certainly more beneficial than Sodium. The Ammonium based product would probably have its uses for the nitrogen component and seeing that humates are used as soil conditioners I would not be surprised if prilled ADA type nutrated plant soils are fortified with this, so I reckon it's has a more targeted use wheras a Potassium based Humic Acid can be applied more liberally as a Tonic in an aquatic environment.
The RAW Full Up product doesn't state its Fulvic Acid content but it does state its Humic Acid content at 10%.

It is stated that it is sourced from Peat so the product should contain a high percentage of Fulvic Acid and other Humates (there's a multitude of different ones, Fulvic and Humic are the big two).
The 10% Humic Acid content is either what's left in the purification process or added purposefully as apparently the presence of Humic Acid can be seen to increase the uptake of Fulvic Acid, possibly because as the plant is uptaking the larger molecule it's holding the door wide open in places (like squeezing a square peg into a round hole, or vice versa) and as consequence the smaller molecules of Fulvic Acid can squeeze through the gaps. So the Fulvic source you found at 62% is likely a very similar product (maybe a different source), it will have other soluble Humates and possibly some undisclosed Humic Acid (don't know how rigorous centrifuging needs to be to separate precipitated Humic Acid from liquid containing Fulvic Acid to have a Zero level for Humic Acid). A combination of both is probably more desirable than Fulvic Acid on its own.
I can't be certain but I dont think there is much of a need to worry too much about harmful contaminants as the original source for these products (excepting possibly the type of Hydroxide pH moderator used to solvate the Humic Acid) as Peat and Leonardite are mainly composed of pre-fossilised plant derived organic matter, so if there are any undesirable contaminants present hopefully they will be at an extremely low level.
Certainly all this DIY malarkey is not for the everyone but I think where there is the absence of an easy to find nutrient source pre-made or one that comes with the required level of desired elements, then learning how to be able to tailor universal base ingredients together in a way to be advantageous to plant health has to be worth the effort.
It's quite interesting to compare the size difference between the Humic and Fulvic Acid molecules. Shortcrust pastry or Puff pastry, I assume they both appear quite delicious from a plants perspective.

This is quite an interesting article - Humates and Humic Acid. How do they work?
Commenting on the purity of the Humic and Fulvic Acid sources you found it might be worth comparing.
Here's the back label for the RAW brand Humic Acid source I mentioned above.

As you can see it's sourced from Leonardite and it is only 59% purity, so your 80% purity grade appears to be a more concentrated product, very likely it is from a locally sourced Leonardite deposit, the question you need to ask is how it was prepared, which hydroxide was used to solvate the Humic Acid, the RAW one above uses Potassium Hydroxide hence the 4% K content, other hydroxides that can be used are Sodium Hydroxide or Ammonium Hydroxide (from what I've read the Ammonium formulation smells pretty bad). I would prefer the Potassium based product simply because it's the preferred transport ion within plants, certainly more beneficial than Sodium. The Ammonium based product would probably have its uses for the nitrogen component and seeing that humates are used as soil conditioners I would not be surprised if prilled ADA type nutrated plant soils are fortified with this, so I reckon it's has a more targeted use wheras a Potassium based Humic Acid can be applied more liberally as a Tonic in an aquatic environment.
The RAW Full Up product doesn't state its Fulvic Acid content but it does state its Humic Acid content at 10%.

It is stated that it is sourced from Peat so the product should contain a high percentage of Fulvic Acid and other Humates (there's a multitude of different ones, Fulvic and Humic are the big two).
The 10% Humic Acid content is either what's left in the purification process or added purposefully as apparently the presence of Humic Acid can be seen to increase the uptake of Fulvic Acid, possibly because as the plant is uptaking the larger molecule it's holding the door wide open in places (like squeezing a square peg into a round hole, or vice versa) and as consequence the smaller molecules of Fulvic Acid can squeeze through the gaps. So the Fulvic source you found at 62% is likely a very similar product (maybe a different source), it will have other soluble Humates and possibly some undisclosed Humic Acid (don't know how rigorous centrifuging needs to be to separate precipitated Humic Acid from liquid containing Fulvic Acid to have a Zero level for Humic Acid). A combination of both is probably more desirable than Fulvic Acid on its own.
I can't be certain but I dont think there is much of a need to worry too much about harmful contaminants as the original source for these products (excepting possibly the type of Hydroxide pH moderator used to solvate the Humic Acid) as Peat and Leonardite are mainly composed of pre-fossilised plant derived organic matter, so if there are any undesirable contaminants present hopefully they will be at an extremely low level.
Certainly all this DIY malarkey is not for the everyone but I think where there is the absence of an easy to find nutrient source pre-made or one that comes with the required level of desired elements, then learning how to be able to tailor universal base ingredients together in a way to be advantageous to plant health has to be worth the effort.
I was actually writing a post for you and you came with all this info. Needless to say all I was writing now goes to the trash as you answered everything I was asking - lol
I do understand more precisely why you want to use humic and fulvic acids and also why you are using Fe DTPA. It all makes perfect sense.
I will try to find out more about the process but I am afraid I wont get any information from the sellers I contacted because these acids seems to be imported from China (which is my main concern) and so far I haven't been able to find a professional chemical reseller selling those acids locally. Those that I have found don't have a clue about the exact composition of the product they are selling and how it is made. I also found another company selling a <..liquid humic/fulvic acid product..> but it contains traces which is no good specially because the company can't even tell me which traces and in what quantities. He says he imports the concentrate and simply repackages it. I kindly asked him if he could convey my request to the manufacturer but I am no getting my hope high on this.
I totally agree and that's why I am not giving up. If we can come up with a recipe that basically drops as much as possible synthetic chelating agents and where all base ingredients can be sourced individually whitout relying on a premix then that would make this fertilizer the best of all I believe.
I do understand more precisely why you want to use humic and fulvic acids and also why you are using Fe DTPA. It all makes perfect sense.
As you can see it's sourced from Leonardite and it is only 59% purity, so your 80% purity grade appears to be a more concentrated product, very likely it is from a locally sourced Leonardite deposit, the question you need to ask is how it was prepared, which hydroxide was used to solvate the Humic Acid, the RAW one above uses Potassium Hydroxide hence the 4% K content, other hydroxides that can be used are Sodium Hydroxide or Ammonium Hydroxide (from what I've read the Ammonium formulation smells pretty bad). I would prefer the Potassium based product simply because it's the preferred transport ion within plants, certainly more beneficial than Sodium. The Ammonium based product would probably have its uses for the nitrogen component and seeing that humates are used as soil conditioners I would not be surprised if prilled ADA type nutrated plant soils are fortified with this, so I reckon it's has a more targeted use wheras a Potassium based Humic Acid can be applied more liberally as a Tonic in an aquatic environment.
I will try to find out more about the process but I am afraid I wont get any information from the sellers I contacted because these acids seems to be imported from China (which is my main concern) and so far I haven't been able to find a professional chemical reseller selling those acids locally. Those that I have found don't have a clue about the exact composition of the product they are selling and how it is made. I also found another company selling a <..liquid humic/fulvic acid product..> but it contains traces which is no good specially because the company can't even tell me which traces and in what quantities. He says he imports the concentrate and simply repackages it. I kindly asked him if he could convey my request to the manufacturer but I am no getting my hope high on this.
Certainly all this DIY malarkey is not for the everyone but I think where there is the absence of an easy to find nutrient source pre-made or one that comes with the required level of desired elements, then learning how to be able to tailor universal base ingredients together in a way to be advantageous to plant health has to be worth the effort.
I totally agree and that's why I am not giving up. If we can come up with a recipe that basically drops as much as possible synthetic chelating agents and where all base ingredients can be sourced individually whitout relying on a premix then that would make this fertilizer the best of all I believe.
Last edited:
Well well I spoke too fast. I got some answers from one of the seller of humic/fulvic acid.
The 80% humic acid I referred to earlier is actually 70% (and not 80%) made with sodium hydroxide and it solubility seems to depend on wheter it is powder, flakes or crystals. The most soluble is the flake version at 100%.
https://www.knrinter.com/category/12/ฮิวมิค-humic

The one made with potassium hydroxide they have 2 kinds. One is 70% and another is at 55%. Solubility depends as well on the type ranging from 85% to 100% solubility.
https://www.knrinter.com/category/2/ซุปเปอร์-โปรแตสเซียมฮิวเมท
https://www.knrinter.com/category/1/โปรแตสเซียมฮิวเมท-2


They also have this product. It's Humic Fulvate and it's 70% Humic Acide + 15% Fulvic Acid:
https://www.knrinter.com/product/70/ฮิวมิค-ฟูลเวท-ชนิดผง-กระสอบ-25-kg
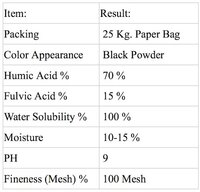
They do also sell Fulvic Acid separately at 50% purity:
https://www.knrinter.com/product/30/ฟูลวิค-ผง-fulvic-25-kg

The 80% humic acid I referred to earlier is actually 70% (and not 80%) made with sodium hydroxide and it solubility seems to depend on wheter it is powder, flakes or crystals. The most soluble is the flake version at 100%.
https://www.knrinter.com/category/12/ฮิวมิค-humic

The one made with potassium hydroxide they have 2 kinds. One is 70% and another is at 55%. Solubility depends as well on the type ranging from 85% to 100% solubility.
https://www.knrinter.com/category/2/ซุปเปอร์-โปรแตสเซียมฮิวเมท
https://www.knrinter.com/category/1/โปรแตสเซียมฮิวเมท-2


They also have this product. It's Humic Fulvate and it's 70% Humic Acide + 15% Fulvic Acid:
https://www.knrinter.com/product/70/ฮิวมิค-ฟูลเวท-ชนิดผง-กระสอบ-25-kg

They do also sell Fulvic Acid separately at 50% purity:
https://www.knrinter.com/product/30/ฟูลวิค-ผง-fulvic-25-kg

Last edited:
Hi all,
The level of impurities in the tech grade will still be vanishingly small. The way I think about is in terms of "lorry loads". So using that as an analogy, the lab. (analytical) grade has a grain of sugar in a lorry load of salt, and the tech. grade a sugar cube in a lorry load of salt.
cheers Darrel
You can use the tech. grade, the lab. grade is for analytical work (like making up standards) and is ultrapure.The lab grades are just insanely expensive.
The level of impurities in the tech grade will still be vanishingly small. The way I think about is in terms of "lorry loads". So using that as an analogy, the lab. (analytical) grade has a grain of sugar in a lorry load of salt, and the tech. grade a sugar cube in a lorry load of salt.
cheers Darrel
@X3NiTH So what do you recommend I do regarding the humic/fulvic acids? Also I have been trying to souce the Fe Gluconate locally and for some reason I haven't found a single supplier. None of the companies I purchased the traces from or those that I enquired have Fe Gluconate. Only sources I found are from ebay and one doesn't ship to Thailand. The other one hasn’t answered. Pharmacies do have Fe Gluconate but it's not pure and pills contain some other ingredients so that's no good.
https://www.ebay.com/itm/BulkSupple...hash=item2f335d3c26:m:mrI7ViDpvW2Kt2Ew0lrJC0Q
https://www.ebay.com/itm/FERROUS-Gl...hash=item2ce89732b7:m:mIEoYScXGLSBtudDDc2ncsA
What are my options at this point? Only use FE DTPA 7% while keep searching for the above?
Your advice and guidance would be appreciated.
https://www.ebay.com/itm/BulkSupple...hash=item2f335d3c26:m:mrI7ViDpvW2Kt2Ew0lrJC0Q
https://www.ebay.com/itm/FERROUS-Gl...hash=item2ce89732b7:m:mIEoYScXGLSBtudDDc2ncsA
What are my options at this point? Only use FE DTPA 7% while keep searching for the above?
Your advice and guidance would be appreciated.
Last edited:
Hi all,
If you really want to add humic, or fulvic, compounds as well you can just add some <"Terminalia catappa"> leaves to the tank.
I like structural leaf litter for all <"sorts of reasons">.
cheers Darrrel
I also bought C6H8O6 (Ascorbic Acid). Would this be ok to replace the Gluconated Iron and instead increase the dose of either Fe-DTPA 7% or Fe-EDTA 13.5%?
You only need one iron (Fe) source, FeDTPA is good for all the pH levels you would encounter in the tank.Only use FE DTPA 7%
If you really want to add humic, or fulvic, compounds as well you can just add some <"Terminalia catappa"> leaves to the tank.
I like structural leaf litter for all <"sorts of reasons">.
cheers Darrrel
Thank you Darell. That clear things up. I guess the intention of @X3NiTH was to reduce as much as possible any synthetic chelating agent. I will try to make a first batch of fertilizer without humic acids and investigate further how to make a solution with said acids. I will do as you said earlier with 3 solutions and only mix them in a 1 month worth all-in-on solution. Should I add ascorbic acide and potassium sorbate to all 3 solutions?
Last edited:
Hi all,
If you have soft water I suspect that citric acid would do as your iron chelator.
They work synergistically, the lower pH from the acid causes the potassium sorbate to convert to sorbic acid and that is what inhibits micro-organism growth.
cheers Darrel
Before the development of FeEDTA people used citric acid (C6H8O7) as their acidifier/chelator and ferrous sulphate (FeSO4.7H2O) as their iron source for hydroponics. I would guess that works about as well as ferric gluconate, and they are both considerably less effective than FeDTPA etc.as much as possible any synthetic chelating agent
If you have soft water I suspect that citric acid would do as your iron chelator.
I can see a reason for adding manganese to the chelator, because manganese and iron deficiency both cause similar looking chlorosis in new growth, and EDTA etc. are very effective chelators and will bind the next available ion (dependent upon frequency and valence).You can also get iron chelates with added manganese, I've bought <"Chempak sequestered iron"> in Wilkos etc. .
Probably just in the "all-in-one", you only need a very small amount of each.Should I add ascorbic acide and potassium sorbate to all 3 solutions? I will do as you said earlier with 3 solutions and only mix them in a 1 month worth all-in-on solution
They work synergistically, the lower pH from the acid causes the potassium sorbate to convert to sorbic acid and that is what inhibits micro-organism growth.
cheers Darrel




